
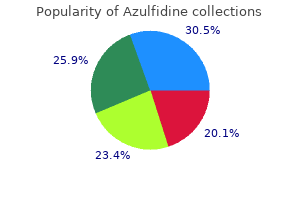
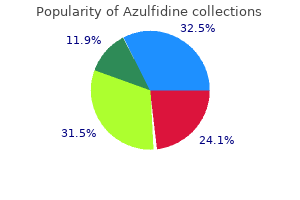
Azulfidine
| Contato
Página Inicial
Ali Bydon, M.D.
- Co-Director of Neurosurgery Medical Student Education
- Professor of Neurosurgery
https://www.hopkinsmedicine.org/profiles/results/directory/profile/0021334/ali-bydon
Of course pain management for dying dog azulfidine 500 mg purchase amex, only erythrocytes can provide oxygen-carrying capacity through hemoglobin pain treatment wellness center 500 mg azulfidine order with amex. Regardless of the volume expander employed stomach pain treatment home purchase azulfidine online, treatment with any volume expander will lead to considerable peripheral edema ocean view pain treatment center generic azulfidine 500 mg free shipping. However davis pain treatment center statesville nc order azulfidine 500 mg with mastercard, the benefits of an increased cardiac output far outweigh the problems associated with peripheral edema. Examination reveals a low blood pressure and tachycardia consistent with low cardiac output. Congestive heart failure Edema Excessive fluid loss in the stool Internal hemorrhage Renal failure Examination shows a very low blood pressure (80/40 mm Hg), tachycardia, a very weak thready pulse, a distended abdomen, and clammy skin. He is diagnosed as having internal hemorrhage leading to severe hypovolemia and circulatory shock. To avoid having the patient go into irreversible shock, the emergency room doctor immediately should initiate which of the following treatments Administration of colloid-free volume expanders (eg, normal saline or lactated Ringer solution) B. His initial blood pressure is 90/60 mm Hg, and the heart rate is 120 beats per minute. On resuscitation with intravenous lactated Ringer solution, his blood pressure increases to 110/70 mm Hg. Two hours later, he is noted to have significant peripheral edema of the hands and feet. Capillary leakage High-output congestive heart failure Infiltration of the intravenous line through the vein Low oncotic pressure Answers [51. Diarrhea over several days can lead to dehydration from loss of fluid in the stool. In severe cases, the individual can become volumedepleted to the point of circulatory collapse. The reduced blood volume and the fall in mean arterial pressure will be sensed by both low-pressure receptors (volume receptors in the atria, pulmonary veins) and high-pressure baroreceptors (carotid, aortic, and afferent arteriole baroreceptors), inducing increased sympathetic nervous activity. This leads to an increase in heart rate, cardiac contractility, and venoconstriction that will serve to elevate mean arterial pressure. All responses to the hypovolemia represent an attempt to return extracellular volume toward normal. The best immediate therapy for a person in hemorrhagic shock is usually isotonic crystalloid colloid-free solution such as normal saline, until red blood cells are available. These agents are usually stocked immediately in the emergency center, whereas blood products require the blood bank to ensure matching blood type. The infusion will increase vascular volume and restore hemodynamics to near normal. Crystalloid such as normal saline cannot restore the hematocrit, but a patient normally can withstand a decrease in hematocrit of up to 20% or so without serious consequences. The use of vasoconstrictors and oxygen can be helpful, but again, if the volume depletion is severe, replacement of fluids will be essential to avoid having the patient go into irreversible shock. Diffuse capillary leakage is the primary reason for the peripheral edema that occurs regardless of which resuscitation fluid is used. The primary defect in circulatory shock is inadequate cardiac output, not just a fall in mean arterial pressure. Blood volume expanders can be used to treat circulatory shock, but only if the patient has not reached the irreversible phase of shock. Increasingly, the understanding of cell and organ function plays an important role in the understanding of disease processes and the treatment of disease. Initially, some of the "language" must be memorized in the same way that the alphabet must be learned by rote; however, the appreciation of the way the "pathology words" are constructed requires an understanding of mechanisms, in essence, an awareness of "how things are put together and work together. The conglomeration of the history, physical examination, and laboratory tests is called the clinical database. After a diagnosis has been reached, a treatment plan is usually initiated, and the patient is followed for a clinical response. Rational understanding of disease and plans for treatment are best acquired by learning about the normal human processes on a basic science level, and likewise, being aware of how disease alters the normal physiologic processes is understood on a basic science level. Given histologic findings in an organ, what are the most likely clinical manifestations Given clinical symptoms, if a tissue biopsy is taken, what histologic findings are most likely to be seen Given clinical findings, if the microscopic photograph is shown, what is the most likely diagnosis Given a gross description of a pathologic lesion, what is the most likely diagnosis This is a fundamental principle in the understanding of the discipline of pathology. The student first must understand the normal histologic structure in an organ in the context of its function. Then the student must be able to relate the abnormal histology to clinical findings, both subjective (patient complaints) and objective (physical examination findings). The organ or system is highly organized both on the gross and on the microscopic level. There also must be awareness of the mechanism that causes disruption of the normal cellular architecture. This is the converse of the first question and requires going backward from clinical manifestations to the probable disease process to probable histologic findings. The student must be able to translate the clinical picture to the cellular characteristics. This also requires being aware of what symptoms various cellular alterations will produce in the patient; for instance, some changes will be silent and not cause symptoms, whereas other changes will produce dramatic manifestations. Given clinical findings, if the microscopic pictograph is shown, what is the most likely diagnosis This sequence of analysis is very similar to the practice of "real-life" medicine, the role of the pathologist. The clinical history and physical examination are critical to putting the pathologic findings into context. For instance, if endometrial curettings are sent to the pathologist and on microscopy reveal crowded, complex glands, abnormal epithelial nuclei, and loss of nuclear polarity, the pathologist may render a diagnosis of cancer. However, when the information is given that the patient is 6week pregnant, the diagnosis of an Arias-Stella reaction is made, an expected finding in the endometrium in light of the human chorionic gonadotropin levels of pregnancy. Thus, the student should be able to shift back and forth between the basic science and the clinical areas Pathophysiology Histologic findings Diagnoses Treatment 4. Given a histologic description, what would be the most likely complication to the organ in question This analysis requires that the student be able to relate the histologic findings of one organ to a disease process and then extrapolate the probable changes to another organ. The student should become proficient at working back and forth between histologic changes and clinical findings and disease processes. The best way to acquire this skill is to think in terms of mechanisms of disease and not just memorize key words. It is the understanding of the underlying pathophysiology of the disease that allows the physician-scientist to make rational predictions of the natural history of a disease process. The student of pathology also must be able to process the visual picture of the organ, biopsy specimen, or cytology, as well as the written description. Because the pathologist often communicates with clinicians by using written reports, the student should be able to take the written description and apply that information to the clinical setting, such as making a diagnosis. For instance, if the description is that of an ovarian cyst with sebaceous material, hair, and teeth, the most likely diagnosis is a benign cystic teratoma. This question is similar to the analysis performed by working back from gross pathologic description to the diagnosis. The student of pathology must be able to correlate the postmortem examination with the probable diagnosis and be able to speculate about the interaction between disease and host. For example, the case may involve a 30year-old female who suddenly collapses and dies, and the autopsy reveals a dilated aortic root and aortic dissection; other findings include long extremities and long fingers. The student once again is challenged to relate the histologic findings in the context of scientific explanation and not just memorize the histologic findings of a certain disease. For example, the histologic specimen may reveal a pulmonary lesion with an area of central necrosis surrounded by epithelioid and multinucleated giant cells. The explanation would be that the organism is probably Mycobacterium tuberculosis, which evades phagocytosis from macrophages because it has complement C3b antigen on its cell wall. It is incorporated into the macrophage, and the tuberculosis bacterium blocks fusion of the lysosome with the phagosome, allowing the bacterium to multiply within the macrophage. Thus, it is delayed or cell-mediated immunity that is required to address the infection. A skilled clinician must be able to translate back and forth between the basic sciences and the clinical sciences. An upper endoscopic examination that was performed recently revealed some reddish discoloration and friability of the lower esophageal region. A biopsy of the lower esophagus was performed, and the microscopic examination revealed columnar cells containing goblet cells. Upper endoscopy reveals reddish discoloration of the distal esophagus, which on biopsy shows columnar epithelium with goblet cells. Most likely mechanism: Repeated acid reflux to the distal esophagus leading to metaplasia of the normal squamous epithelium into columnar epithelium. Through a poorly understood mechanism, the lower esophagus changes (metaplasia) from squamous to columnar epithelium, so-called Barrett esophagus. In fact, the presence of goblet cells in the columnar epithelium is a hallmark of the disease. Barrett esophagus appears reddish and friable on endoscopy and carries an increased risk for developing into adenocarcinoma. Definitions Esophageal diverticulum: Outpouching of one or more layers of the esophageal wall. When it occurs near the upper esophageal sphincter, it is called a Zenker diverticulum. Achalasia: Condition of esophageal dilation resulting from lack of esophageal peristalsis and constant contraction of the lower esophageal sphincter associated with a loss of myenteric plexus ganglions. Barrett esophagus: Columnar metaplasia of the lower esophageal epithelium, predisposing to esophageal adenocarcinoma. Esophageal carcinoma: Worldwide, squamous cell carcinoma is the most common cell type, but in Western countries, it is divided equally in frequency between adenocarcinoma and squamous cell carcinoma. It has a well-developed submucosa, and the upper third is enveloped by striated muscle, whereas the lower twothirds are encompassed by smooth muscle. The upper esophageal sphincter is located at approximately the level of the fifth cervical vertebra (C5) level, whereas the lower esophageal sphincter is located below the diaphragm and its function is to prevent regurgitation of gastric acid. Both sympathetic and parasympathetic nerve fibers innervate the intrinsic myenteric plexus, which is distributed in the striated and smooth muscle. The most common type (90 percent) involves distal esophageal atresia with a connection to the trachea. Maternal polyhydramnios may be noted in utero, resulting from the fetal inability to swallow amniotic fluid. This nearly always involves a loss of myenteric ganglion cells, although the underlying etiology is unclear. By far, the most common cause of esophagitis is gastroesophageal reflux, which may be associated with a hiatal hernia. With chronic acid exposure, the distal esophagus may become hyperemic and ulcerated and develop scars or strictures. Barrett esophagus appears reddish and friable on endoscopy and is diagnosed by biopsy; endoscopic surveillance is important because of the increased risk for developing adenocarcinoma. Esophageal Carcinoma Esophageal cancers account for about 10 percent of all gastrointestinal cancers in the United States and are largely asymptomatic. Chronic alcohol and tobacco exposures significantly increase the risk of esophageal cancer. Other factors may include ingestion of nitrosamine-containing foods, chronic hot and spicy foods, and lye with stricture formation. The most common cell type worldwide is squamous cell carcinoma, usually affecting the upper and middle thirds of the esophagus. Regardless of cell type, affected patients generally have few symptoms until late in the course, with those symptoms being progressive dysphagia, weight loss, and fatigue. Because the cancers are usually very large at diagnosis, surgical resection is difficult, and up to 80 percent of affected individuals die within 1 year of diagnosis. He denies ingestion of unusual substances and complains of pain even when swallowing liquids. Brown blotches scattered throughout the esophagus Normal-appearing esophagus Red patches in the distal esophagus Reddish streaks throughout the pharynx and upper esophagus White patches adherent to the esophagus [1. A pediatric feeding tube is passed orally to 20 cm without difficulty, with gastric secretions aspirated. Congenital heart disease Floppy epiglottis Respiratory distress syndrome Tracheoesophageal fistula Zenker diverticulum Answers [1. Adenocarcinoma is the most common malignancy of the lower third of the esophagus and is strongly associated with Barrett esophagus. Squamous cell carcinoma is the most common type of cancer of the esophagus worldwide and usually affects the upper or middle region of the esophagus. When the baby feeds, the formula is aspirated into the tracheobronchial tree, leading to choking and cyanosis. This condition may be diagnosed with a radiologic contrast study and requires surgical correction. Gastric acid reflux into the distal esophagus may cause esophagitis, and a patient with gastroesophageal reflux disease typically complains of heartburn. He takes approximately one 500-mg acetaminophen tablet a week for headaches but does not take any other medications. There is mild edema in the adjacent mucosa, but there is no thickening of the edges of the ulcer. Long-term complications: Erosion or perforation with bleeding; gastric carcinoma in patients with chronic gastritis. Most likely mechanism: Most often associated with Helicobacter pylori organisms that produce bacterial urease and protease, damaging the mucus layer and exposing the underlying epithelium to acid-peptic injury. Peptic ulcers are often remitting, relapsing lesions that may be seen in young adults but more often occur in middle-aged to older adults. They are usually chronic, solitary lesions caused by the action of gastric acid and pepsin, both of which are thought to be required for the development of peptic ulcers.
Schematic diagram showing the central role of glutamine as a transporter of amino acid nitrogen to various tissues pain management dogs cats 500 mg azulfidine buy with visa. Conditions that influence the tryptophan-to-branched-chain amino acid ratio in the circulation have been shown to affect serotonin levels in the brain pain treatment for bursitis buy azulfidine 500 mg fast delivery, because of competition for this transporter natural pain treatment for dogs generic 500 mg azulfidine free shipping. For example brunswick pain treatment center purchase azulfidine with visa, increased skeletal muscle branched-chain amino acid catabolism during starvation is associated with an increase in the blood tryptophan-to-branched-chain amino acid ratio kearney pain treatment center buy azulfidine 500 mg low cost, increased tryptophan uptake in the brain, and increased serotonin synthesis. The guanidino group from arginine is used in the synthesis of nitric oxide, a ubiquitous, highly reactive signaling molecule that regulates multiple physiologic processes, including blood pressure, immune response, learning, and metabolism. Much less understood is the mechanism by which leucine affects cellular signaling. This amino acid has been termed a "pseudohormone" following the observations that one or more of its early metabolites. A 12-year-old girl is presented at the clinic with reports of frequent fainting and lethargy. Proteinamino acids Glucosefatty acids Glucoseglycogen Fatty acidstriacylglycerol Answers [39. In contrast, decreased insulin levels associated with type I diabetes, anorexia, and bulimia, will promote lipolysis and therefore decreased adipose mass. The low body weight and fat mass observed in the patient are consistent with a metabolically "fasted" state. During such a condition, circulating insulin levels will be low, whereas counterregulatory hormones. Consistent with a metabolically fasted state, decreased circulating insulin levels will signal attenuation of biosynthetic reaction and augmentation of catabolic reactions. The latter will provide necessary fuels to sustain the energetic needs of the body. Increased proteolysis (proteinamino acids) in tissues such as skeletal muscle and the liver provide amino acids as a direct fuel source, as well as ketogenic and gluconeogenic precursors (depending on the individual amino acid). Amino acids can be divided into one of two classes, essential and nonessential amino acids. Nitrogen balance means whether there is more or less nitrogen (protein) ingested than excreted. With prolonged caloric insufficiency, a large portion of the carbon skeleton is used by the liver for the synthesis of either glucose or ketone bodies, depending on the specific amino acid. Her friends were concerned when she did not show up for a biochemistry final at the local university. The patient had been under a lot of stress with finals, a recent breakup with a boyfriend, and trying to find a job. In the dormitory room, one of her friends noticed an empty bottle of Tylenol (acetaminophen) near the bed with numerous pills lying on the ground near their friend. On arrival to the emergency department, the patient was found to be in moderate distress and vomiting. Her urine drug screen was negative, and her acetaminophen blood level was above 200 g/mL. The emergency department physician prescribes oral N-acetylcysteine to help prevent toxicity from the acetaminophen. What is the biochemical mechanism whereby the N-acetylcysteine helps in this condition Because the liver has high levels of cytochrome P450 enzymes, it is the major organ affected by acetaminophen overdose. Biochemical mechanism of N-acetylcysteine: As glutathione is used to conjugate the acetaminophen toxic metabolite, the antidote Nacetylcysteine helps to facilitate glutathione synthesis by increasing the concentrations of one of the reactants of the first synthetic step. Normally acetaminophen is cleared by conjugation with either glucuronic acid or sulfate followed by excretion. Metabolism also takes place, producing an active intermediate capable of binding tissue macromolecules. These conjugative and metabolic pathways involve a number of enzymes that may themselves be compromised to such an extent that the threshold for the concentration that constitutes an overdose is substantially lowered. More typically, overdose concentrations are the result of deliberate ingestion, as in this clinical case, or accidental ingestion, often involving either a child who finds a bottle of acetaminophen and consumes its contents or a disoriented elderly person who loses track of how many tablets have been consumed. Usually, the acetaminophen serum level is drawn and plotted on a nomogram to determine the possibility of hepatic damage. Hepatocyte necrosis with clinical manifestations of nausea and vomiting, diarrhea, abdominal pain, and shock may ensue. The initial therapy is gastric lavage, activated charcoal, supportive care, and administration of N-acetylcysteine. Be aware of the mechanism of action of N-acetylcysteine in treating acetaminophen toxicity. Definitions Phase I drug metabolism: Oxidative metabolism of drugs usually mediated by cytochrome P450 leading to hydroxylation or epoxidation of substrate compounds. Drug toxicity: Aberrant reaction to a therapeutic agent often depending on individual variations in either the quantity or activity level of specific drug metabolizing enzymes toward the drugs or on individual genetic polymorphisms of drug metabolism enzymes giving higher or lower activities or product profile produced by the genetic variant versus the normally expressed enzyme. Phosphoglucomutase, which plays a critical role in glycogen formation, converts glucose 6-phosphate to glucose 1-phosphate. Alternatively, acetaminophen can be conjugated with organic sulfate for elimination. In the event that this pathway and the glucuronide forming pathway are both overwhelmed by overdose, more acetaminophen is metabolized by the cytochrome P450 pathway. The tissue with the highest concentration of cytochromes P450 is the liver, and it is there (along with kidney) that acetaminophen causes the most damage. However, N-acetyl benzoquinoneimine may also disrupt Ca2+ balance, leading to dramatically increased intracellular Ca2+ concentrations, that is, 20 M versus 0. Thus dramatic increases in Ca2+ concentration would have deleterious effects on the balance of many cellular processes, especially energy generation. The low serum calcium and elevated liver enzyme serum levels seen in the patient reflect hepatocyte lysis. The adduct acetaminophen glutathionate is no longer toxic to cells and can be excreted without further damage. Any process depleting glutathione levels would compromise the ability of the cell to protect itself against N-acetyl benzoquinoneimine. This includes deficiencies of any of the enzymes involved in the synthesis of glutathione or that keep glutathione in its reduced state as well as any other radical generating process that consumes glutathione. In the case of acetaminophen overdose, in addition to giving activated charcoal to absorb excess stomach and intestinal acetaminophen or its conjugates, a strategy to replenish glutathione concentration is important. Easily obtainable and readily soluble, it serves to facilitate glutathione synthesis by increasing the concentrations of one of the reactants of the first synthetic step. The committed dipeptide product being made, glutathione is synthesized to replenish depleted supplies. N-acetylcysteine seems most effective when given less than 10 hours after ingestion of acetaminophen but is recommended within the first 35 hours after ingestion. What is the most probable explanation of how a high dose of acetaminophen might have led to a toxic condition Acetaminophen is metabolized to a potential toxic product that is fully conjugated. Acetaminophen is metabolized to a potential toxic product that is partially conjugated. Acetaminophen glucuronide Acetaminophen sulfate N-Acetyl benzoquinoneimine Acetaminophen glutathionate N-Acetyl-p-aminophenol [40. Glutamic acid, alanine, methionine Glutamine, alanine, cysteine Glutamate, glycine, cysteine Alanine, glycine, cysteine Methionine, glycine, cysteine Answers [40. Acetaminophen is not itself toxic, but its intermediate metabolite N-acetyl benzoquinoneimine can be toxic unless it is adequately conjugated. In the case presented, a high dose of acetaminophen overwhelmed the conjugative processes, allowing the toxic intermediate to interact with body components and thus causing the nausea, vomiting, and elevated blood enzymes observed. N-acetyl benzoquinoneimine is toxic, whereas acetaminophen glucuronide, acetaminophen sulfate, and acetaminophen glutathionate are nontoxic acetaminophen conjugates. Glutathione (-glutamylcysteinylglycine) is a tripeptide of glutamic acid, cysteine, and glycine in which the amino terminal glutamate residue is in a peptide linkage through its side-chain carboxyl group to the cysteine residue. Acetaminophen is oxidized by the cytochrome P450 system, yielding the deleterious product N-acetyl benzoquinoneimine, an unstable intermediate that can react with cellular macromolecules and thus damage them. The liver has a high concentration of cytochrome P450 and is particularly susceptible to acetaminophen toxicity. N-acetylcysteine is the antidote to acetaminophen toxicity and facilitates glutathione synthesis by increasing the concentrations of one of the reactants of the first synthetic step. The patient heard from a friend about a new vegetarian diet that promised rapid weight loss. The diet consists of many leafy vegetables with no pork, chicken, beef, eggs, or milk. She is also planning on working out regularly with the goal of running a marathon within the year. After listening to the patient, you refer her to a nutritionist for further assistance and guidance. List of essential amino acids: Histidine, isoleucine, leucine, lysine, methionine, phenylalanine, threonine, tryptophan, and valine. Most animal proteins contain all the essential amino acids; however, vegetable proteins often lack one or more of them. Vegans often benefit from nutritional consultation, which would allow the patient to make sure that she ate foods that complemented each other in providing the essential amino acids. Definitions Essential amino acids: Amino acids that the human body cannot synthesize (or cannot synthesize in sufficient quantities to meet cellular needs) and must be taken in the diet. If essential amino acids are deficient, the result is a condition of negative nitrogen balance. Nonessential amino acids: Those amino acids that are synthesized by the human body in sufficient quantities to meet cellular needs. Phenylalanine hydroxylase: the enzyme that converts the essential amino acid phenylalanine to the amino acid tyrosine using tetrahydrobiopterin and molecular oxygen. These metabolites are elevated when the conversion of phenylalanine to tyrosine is impaired. Amino acids are the basic building blocks for all proteins synthesized in the cell. In addition, the metabolism of amino acids provides carbon and nitrogen units that are used in the synthesis of numerous important biomolecules including neurotransmitters, heme, purines, pyrimidines, polyamines, and various cellularsignaling molecules. Apart from their biologic importance, amino acids are classified as essential or nonessential based on their ability to be synthesized in the body. The term essential amino acid is used to identify those amino acids that must be taken in through the diet (Table 41-1). There are 10 such amino acids for which biosynthetic pathways do not exist in cells of the human body. In contrast, there are 11 amino acids that are termed nonessential, for which the human body has biosynthetic pathways for their generation. In addition, it should be noted that arginine is listed as both an essential and nonessential amino acid. Arginine is considered nonessential because biosynthetic pathways for its generation do exist in certain cells of the body. The urea cycle is found only in the liver, and thus the production of arginine through this pathway is limited. The production of arginine through this pathway is likely sufficient for healthy adults but may not be sufficient in times of growth when increased protein synthesis augments the need for amino acids. Thus, in growing children and in adults following surgery or trauma, arginine becomes an essential amino acid. The most readily available source of all amino acids, but particularly the essential amino acids, is the diet. Protein is taken in through the diet and digested to smaller peptides and amino acids in the stomach and small intestine by specific proteolytic enzymes known as proteases. Because of their different specificities, enzymes work to cleave specific peptide bonds within proteins. Individual digestive enzymes are not capable of completely digesting proteins themselves, but in concert with many different enzymes, most proteins can be efficiently digested. Once released by the digestive enzymes, amino acids are absorbed by epithelial cells of the small intestine for distribution and utilization throughout the body. Composition of the diet is an important consideration when trying to understand and plan for the uptake of essential amino acids. Not all dietary constituents are equal with regard to the type and amounts of proteins present or the amino acids that can be derived from these proteins by the human digestive tract. Proteins derived from vegetable matter may not contain all the essential amino acids needed and digestion of certain plant proteins can be insufficient to produce certain individual amino acids. In contrast, proteins found in animal products are readily digestible and contain all essential amino acids. Therefore, careful consideration should be given to the dietary intake of individuals who may have or will be undergoing an increased level of exertion. Phenylalanine is an essential amino acid that is also used to generate the nonessential amino acid tyrosine. Diets low in phenylalanine will help prevent excessive elevations in phenylalanine. Phenylalanine can not be completely eliminated from the diet because it is an essential amino acid needed for protein synthesis. Which of the following amino acids is an essential amino acid under conditions of enhanced growth or surgical recovery Fortunately, this condition can be managed by regulating the amount of phenylalanine provided in the diet. Which of the following nonessential amino acids will need to be supplied in the diet of this infant Methionine can be synthesized by the methylation of homocysteine by the enzyme methionine synthase, which requires the participation of vitamin B12 and 5-methyltetrahydrofolate. It is actually the homocysteine component of methionine that is required, since this reaction has the capacity to synthesize enough methionine. Glutamate is first converted to ornithine, which is then converted to arginine by enzymes of the urea cycle.
It catalyzes the hydrolysis of mono- pain treatment west plains mo azulfidine 500 mg purchase overnight delivery, di- pain center treatment for fibromyalgia trusted azulfidine 500 mg, and triglycerides as well as the phospholipids phosphatidyl choline and phosphatidyl ethanolamine laser pain treatment reviews cheap azulfidine 500 mg fast delivery. Chylomicrons are formed in the epithelial cells of the intestine and are responsible for the transport of dietary lipids the pain treatment and wellness center azulfidine 500 mg buy overnight delivery. Dietary triglycerides are hydrolyzed by pancreatic lipase in the lumen of the small intestine dental pain treatment guidelines order 500 mg azulfidine overnight delivery. The free fatty acids and monoglycerides are absorbed by the microvilli of the intestinal epithelial cells. In the epithelial cell, the fatty acids and monoglycerides are reformed into triglycerides, which are packaged with phospholipids, cholesterol, and apolipoprotein B-48 into chylomicrons. The newly synthesized chylomicrons are secreted into the lymph and enter the bloodstream via the thoracic duct. The fatty acids are taken up by the adipose or muscle cells; glycerol is recycled back to the liver. The remnants are taken into the hepatocytes by endocytosis and degraded in the lysosome to fatty acids, amino acids, cholesterol, glycerol, and phosphate. Chylomicrons appear in the blood stream shortly after consumption of a meal containing fat. However, the clearance rate for chylomicrons is fast and blood is usually free of chylomicrons following an overnight fast. Hypertriglyceridemia can be the result of a genetic disorder in one of the proteins involved in lipoprotein metabolism, or it can arise secondarily to a number of other disorders, including diabetes mellitus, obesity, and alcohol abuse, and as a side effect of some medications such as -blockers, oral estrogens, and some diuretics. Decreased lipoprotein lipase activity is the result of the failure of the pancreatic -cells to produce and secrete insulin. Insulin stimulates the synthesis of lipoprotein lipase; in the absence of insulin, lipoprotein lipase activity in the capillary beds is low. If a Type I diabetes mellitus patient is not given insulin, the increased glucagon/insulin ratio would stimulate gluconeogenesis and -oxidation in the liver rather than synthesis of triglycerides. One of the first patients you encounter is an 8-monthold girl brought to the clinic because of excessive exhaustion and fatigue. On further questioning of the mother, she reports that she was previously breastfeeding but had to stop to return to work. To feed all of her other children, she has had to dilute her formula with water to make the formula last longer for the entire family. After your physical exam is performed, you diagnose the infant with severe malnutrition and aid the mother with resources to increase food intake for their household. In marasmus, a child usually between the ages of 1 to 3 years has inadequate caloric intake leading to loss of subcutaneous fat, loose wrinkled skin, and either flat or distended abdomen resulting from atropic abdominal wall muscles. Be familiar with the metabolic change in fasting states as compared to starvation. Definitions Marasmus: Malnutrition resulting from inadequate intake of protein and calories. Kwashiorkor: Malnutrition resulting from inadequate intake of protein though the intake of total calories is adequate. Ketone bodies: the short chain fatty acid metabolites acetoacetate and -hydroxybutyrate and acetone. Triglyceride: A glycerol molecule with each hydroxyl group esterified with a fatty acid moiety. Monoglyceride: A glycerol molecule with one hydroxyl group esterified with a fatty acid moiety. The case description reveals that the child lives in a third-world country, and the physical findings reveal that the child suffers from protein-calorie-deficient starvation, or marasmus. Fasting and starvation represent changes from the baseline metabolic interactions between tissues that exist in the fed state. Each of three states-fed, fasting, and starvation-must be considered from the standpoint of the whole body primarily because the constituent tissues have different requirements for their nutritional sources. For example, red blood cells have an absolute requirement for glucose as the exclusive food source from which energy is derived. Although other tissues use fatty acids and amino acids as well, the red blood cell cannot because it lacks mitochondria and therefore the enzymes required for most of the metabolic steps required in -oxidation of fatty acids and metabolism of the carbon skeletons of amino acids. Brain tissue normally has an exclusive preference for glucose, the exception being in advanced starvation when the brain can use ketone bodies for energy production. Glucose, fatty acids from triglycerides, and amino acids are provided by the diet and used differentially by the tissues. In the liver, glucose is used for storage as glycogen or converted to fatty acids for formation into triglycerides for storage in adipose tissue. Amino acid carbon skeletons are used for metabolic intermediates for energy production or fatty acid synthesis. Resting muscle takes up glucose and stores it as glycogen and uses amino acids for protein synthesis. Resting muscle prefers fatty acids and ketone bodies over glucose to satisfy its energy demands. Glucose metabolism provides energy and glycerol 3-phosphate for triglyceride formation and storage using fatty acids transported to adipose cells as triglycerides in lipoprotein particles. The liver shifts from consumption of glucose for glycogen storage to mobilization of its glycogen stores to release glucose to the bloodstream to supply the glucose requirements of the brain and red blood cell. Increased blood glucose triggers release of insulin and a decrease in the release of glucagon and lipolytic hormones. The energy required for gluconeogenesis is derived by increasing -oxidation of fatty acids mobilized from adipose storage sites. Blood glucose levels begin to decrease, triggering homeostatic mechanisms to prevent it from decreasing dramatically. This occurs only in liver mitochondria because of its critical role in gluconeogenesis. The b-oxidation of fatty acids that occurs in the mitochondrial matrix provides the energy for gluconeogenesis in the liver. Mobilization of fatty acids during times in which the liver is synthesizing glucose via the gluconeogenic pathway. The ketone bodies are exported to extrahepatic tissues, where they are used as an energy source. The fatty acids are transported across the mitochondrial inner membrane as carnitine derivatives utilizing the carnitine shuttle. The activation of fatty acids and transport into the mitochondrion via the carnitine shuttle. The newly produced fatty acylCoA repeats the cycle of steps in -oxidation releasing another acetyl-CoA and onward until the last cleavage step that hydrolyzes acetoacetyl-CoA to two molecules of acetyl-CoA. Thus starvation represents an intensification of the metabolic adjustments of the fasting state with some significant differences seen only in prolonged starvation. The brain adapts to use ketone bodies as a source of energy, thus decreasing its utilization of glucose. Muscle activity decreases as result of the mobilization of muscle protein, which itself slows as the period of starvation increases. In brain and the central nervous system an adaptive change occurs allowing this tissue to use ketone bodies as an energy source relieving both the total body demand for glucose and the use of muscle protein as a carbon source for gluconeogenesis in the liver. Thus the major differences between starvation and the postfeeding fast are the adaptive ability of the brain and central nervous system to use ketone bodies to satisfy some of their energy demand and in the levels of circulating ketone bodies that are high enough to spill over into the urine in significant quantities. The 8-month-old was fed diluted formula, and the tentative diagnosis of marasmus was made. Which of the following illustrates starvation-triggered changes in intermediary metabolism Increased dependence of liver on glucose for its energy supply Increased synthesis of proteins in muscle tissue Increased use of ketone bodies for energy source in brain Decreased mobilization of triglycerides by adipose tissue Adaptation of red blood cells to use ketone bodies for energy Use the reactions below for Question 36. Which of the following amino acids remains in the muscle cell to provide a source of energy for the muscle In starvation a major metabolic adjustment is that the brain activates the ketone body metabolic pathway and uses ketone bodies for energy, thus sparing somewhat the breakdown of body proteins to generate amino acid carbon skeletons for gluconeogenesis in the liver. Triglyceride stores in adipose tissue are being used to provide fatty acids for -oxidation in the liver. Red blood cells are not able to use ketone bodies because they have no mitochondria. The reaction of succinyl-CoA and acetoacetate is a reaction in the pathway of ketone body utilization but not in the pathway of ketone body formation. The muscle has a very active branched-chain amino acid metabolic pathway and uses that pathway to provide energy for its own use. The products of leucine metabolism are acetyl-CoA and acetoacetate, which are used in the tricarboxylic acid cycle. Acetoacetate is activated by succinyl-CoA and cleaved to two molecules of acetyl-CoA in the -ketothiolase reaction. The other branched-chain amino acids, valine, and isoleucine, yield succinylCoA and acetyl-CoA as products of their catabolism. The family reports that the patient has been very disoriented and confused over the last few days and has been nauseated and vomiting blood. The family first noticed disturbances in his sleep pattern followed by alterations in his personality and mood. A blood ammonia level was noted to be elevated, and all other tests have been normal. Nonrhythmic asymmetric tremor with loss of voluntary control of extremities while in a sustained position. The patient usually manifests malaise, lethargy, palmar erythema, ascites, jaundice, and hepatic encephalopathy in the late stages. The most common etiologies of cirrhosis are toxins such as alcohol, viral infections such as hepatitis B or C infection, or metabolic diseases in children (Wilson disease, hemochromatosis, or 1-antitrypsin deficiency). Treatment depends on the exact etiology, although the common therapy includes avoidance of liver toxins, salt restriction, and possibly procedures to reduce the portal pressure. Ornithine: An -amino acid similar in structure to lysine but having one methylene group less in the side chain. It is carbamoylated to form citrulline to begin the urea cycle and is regenerated in the final step that releases urea. Transaminase: An aminotransferase; a pyridoxal phosphate-requiring enzyme that transfers an amino group from an -amino acid to an -keto acid. Urea cycle: the series of reactions that occur in the liver to synthesize urea for the excretion of nitrogen. The two nitrogen atoms present in urea arise from ammonium ion and the -amino group of aspartate. For the carbons in amino acids to enter into the energy generating metabolic pathways, the amino groups must first be removed so that they can be detoxified and excreted. The amino acid nitrogen is excreted predominantly as urea, but some is also excreted as free ammonia in order to buffer the urine. This process is catalyzed by transaminase (aminotransferase) enzymes that require pyridoxal phosphate as a cofactor. The products of this reaction are glutamate (Glu) and the -ketoacid analog of the amino acid destined for catabolic breakdown. The transamination process is freely reversible, and the direction in which the reaction proceeds is dependent on the concentrations of the reactants and products. These reactions do not effect a net removal of amino nitrogen; the amino group is only transferred from one amino acid to another. For net removal of amino nitrogen, a second enzymatic reaction must take place that removes the amino group from Glu for disposal. Ammonia is produced by almost all cells in the body; however, only the liver has the enzymatic machinery to convert it to urea. However, ammonia in the blood is toxic to cells, and therefore the nitrogen from amino acid catabolism is transported in blood either as glutamine or alanine. Glutamine and alanine are transported to the liver in the blood, where they are taken up by cells in the periportal region. Glutaminase catalyzes the hydrolysis of the side-chain amide group releasing ammonia and Glu. The activated carbamoyl group is then transferred to the amino acid ornithine by the mitochondrial enzyme ornithine transcarbamoylase to form citrulline. Argininosuccinate is then cleaved to arginine and fumarate by argininosuccinase (argininosuccinate lyase). Arginase then hydrolyzes the guanidino group of arginine, releasing urea and regenerating ornithine, which can then reenter the mitochondrion and accept another carbamoyl group from carbamoyl phosphate. Glutamine is also used by the kidney as a source of ammonia that is used to buffer the urine. Ammonia is released from glutamine by the same enzymes that are active in the liver. The free ammonia accepts a proton to form ammonium ion, thus decreasing the acidity of the urine. Although most of the ammonia detoxified by the liver arises from the breakdown of amino acids in dietary protein or endogenous protein that is being turned over, ammonia is also produced by bacteria in the gut. This is absorbed into the portal venous blood and taken directly to the liver for conversion into urea. This can be exacerbated by an increase in the ammonia load, such as can occur from gastrointestinal bleeding. His pulse was elevated, his liver was enlarged, and an electroencephalogram was grossly abnormal. Since the infant could not retain milk given by gavage feeding, intravenous glucose was administered. Analysis of his urine showed abnormally high amounts of glutamine and uracil, which suggested a high blood ammonium ion concentration. Arginase Carbamoyl phosphate synthetase I Glutamate dehydrogenase Glutaminase Ornithine transcarbamoylase [37. The patient appeared to be developing normally until 4 months of age, when he developed truncal hypotonia and spasticity of the limbs. Despite being on a low-phenylalanine diet, at 5 months the patient had several grand mal (epileptic) seizures. A low-Phe diet with biopterin supplementation A low-Phe diet with cobalamin (vitamin B12) supplementation A low-Phe diet + L-dopa (3,4-dihydroxyphenylalanine) A low-Phe diet + L-dopa and 5-hydroxytryptophan A diet completely free of Phe Answers [37.
Discount azulfidine 500 mg mastercard. How to pull a broken tooth.
Diseases
- Gamstorp episodic adynamy
- Radial hypoplasia, triphalangeal thumbs and hypospadias
- Gray platelet syndrome
- Nasopharyngitis
- Hyperlipoproteinemia type II
- Minamata disease
- Marion Mayers syndrome
References
- Fielding AK, Rowe JM, Richards SM, et al. Prospective outcome data on 267 unselected adult patients with Philadelphia chromosome-positive acute lymphoblastic leukemia confirms superiority of allogeneic transplantation over chemotherapy in the pre-imatinib era: results from the International ALL Trial MRC UKALLXII/ECOG2993.
- Brandstedt J, Almquist M, Manjer J, et al: Vitamin D, PTH, and calcium in relation to survival following prostate cancer, Cancer Causes Control 27(5):669n677, 2016.
- Becker JA, Vlach J, Raben N, et al. The African origin of the common mutation in African-American patients with glycogen storage disease type II (GSDII). Am J Hum Genet 1998;62:991.
- Soliman PT, Westin SN, Iglesias D, et al. Phase II study of everolimus, letrozole, and metformin in women with advanced/recurrent endometrial cancer. J Clin Oncol 2016:5506.
- Kaplinsky N, Neumann G, Harzahav Y, Frankl O. Esophageal ulceration in Behcet's syndrome. Gastrointest Endosc 1977;23:160.
- Little JA, Cox D, Breckenridge WC, McGuire VM. Introduction to deficiencies of apolipoproteins CII and EIII with some associated clinical findings. In: Gotto Jr AM, Smith LC, Allen B (eds). Atherosclerosis V. New York: Springer-Verlag; 1980, 671.
- Gorelick PB, Rodin MB, Langenberg P, et al. Weekly alcohol consumption, cigarette smoking, and the risk of ischemic stroke: Results of a case-control study at three urban medical centers in Chicago, Illinois. Neurology 1989;39:339.
