
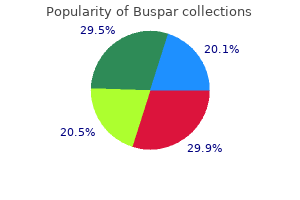
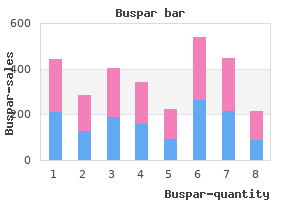
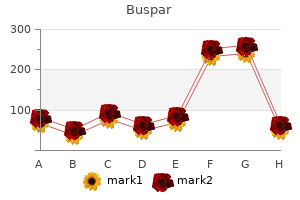
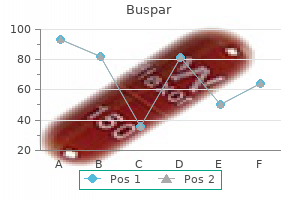
Buspar
| Contato
Página Inicial
Lingye Chen, MD
- Medical Instructor in the Department of Medicine
https://medicine.duke.edu/faculty/lingye-chen-md
However anxiety herbs cheapest generic buspar uk, one typically must examine numerous samples to appreciate the characteristic features of these tumors anxiety symptoms to get xanax discount buspar 10 mg mastercard, which include slit-like spaces or a pseudoglandular pattern anxiety jelly legs buspar 10 mg buy online. Using immunohistochemical techniques anxiety symptoms in teens generic buspar 10 mg without prescription, cells within synovial sarcomas show a biphasic pattern of intermediate filament protein expression anxiety symptoms definition quality buspar 5 mg, with the spindle cells containing vimentin and the plump epithelial-like cells staining for cytokeratin. Fibrosarcomas rarely develop in the periarticular tissues and can invade joints directly. These can be differentiated from synovial sarcomas because fibrosarcomas do not express cytokeratins. It is typically slowly progressive, and may be present at a high incidence microscopically in older animals. When osteoclasts take up bisphosphonates released from mineralized bone and cartilage by acid digestion, their function is impaired. The osteoclast may undergo apoptosis, have a shortened lifespan, and exhibit morphologic changes such as the formation of giant osteoclasts. Secondary Toxicity Since skeletal tissues do not play a major role in biotransformation or elimination of potentially toxic substances from the body, most exogenous chemicals exert their effects on the bone via secondary mechanisms. It has been suggested that secondary toxic effects may be mediated by alterations in blood flow within bones. Increased oxygen tension is thought to stimulate bone resorption locally, while decreased oxygen tension causes bone to accumulate. The close similarity of some drug-induced bone reactions to those observed in spontaneous diseases mediated by a vascular reaction supports the argument for a vascular pathogenic mechanism. Conversely, such reactions could be a manifestation of the limited number of ways in which skeletal tissues can respond to injury. Substances capable of inducing secondary bone changes usually are hormones, vitamins, or minerals, or affect the metabolism of skeletalregulating agents. Agents reported to have a direct toxic effect on bone and/or cartilage are somewhat limited. It may be that we have not yet learned to evaluate the early primary toxic effects of drugs, chemicals, or other environmental agents on hard tissues. A list of agents and toxic mechanisms causing skeletal pathology are given in Table 23. Bone Toxicity Primary Toxicity Aseptic necrosis of the femoral head caused by alcohol consumption or corticosteroid administration is not due to direct drug-induced necrosis of osteocytes, but rather to compromised local circulation in a region of bone that is anatomically vulnerable due to its hemispherical shape and considerable range of motion. In contrast, cyclophosphamide, a cytotoxic antineoplastic agent, directly influences mitotic division of bone cells, thereby inhibiting bone formation and growth. The characteristic regular joint surface from the unaffected left acetabulum (C, E, G) lacks fibrillated cartilage and the loss of cartilage matrix integrity of the damaged right side (D, F, H). There is a wide divergence in the way many agents produce their effects, which can be observed at different organizational levels. Since bone turnover events represent a sequence of different biological activities, intermittent, brief, or continuous delivery of the same agent may give distinct skeletal responses. Skeletal responses to pharmacologic agents may diminish with time as tolerance develops. One probable factor responsible for different drug doses causing varied biological responses is the differential action of drugs on the modeling and turnover stages. For example, a very small dose of 1,25-dihydroxy vitamin D3 reverses the endochondral growth abnormality associated with vitamin D deficiency, but high doses can produce markedly increased osteoid in the metaphysis. It is important to recognize that skeletal cell-to-cell and cell-to-matrix interactions involve both bone cells and cells of the bone marrow and are controlled by both systemic and local factors. Therefore, cell functional activities and cellular interactions represent the final common pathway of toxic action rather than a basic mechanism of toxic action. Local events are influenced by circulating agents such as hormones, but the effects of circulating agents are also determined by local factors such as mechanical usage. In addition, the processes active in bone disease are not necessarily the same processes operating in physiologically normal states. Certain diseases appear to affect one bone envelope (periosteal, Haversian, endosteal) more than others. In general, the endosteal bone envelope is more reactive and responsive than the Haversian envelope because endosteal (endocortical and trabecular) bone possesses a greater cumulative surface area, has cells with higher metabolic activity, and experiences higher bone turnover. The surface-to-volume ratio and turnover rate is three times higher in trabecular bone than cortical bone in both humans and dogs. In the rapidly growing young rat, the endocortical surface of the metaphysis is predominantly formative, while the periosteal metaphyseal surface is predominantly resorptive as the diameter of the bone is progressively reduced from wide at the physis to relatively narrow at the diaphysis. Certain drugs also appear to affect particular envelopes, and there are species differences as well. Mice treated with estrogens produce much more marrow cancellous bone than do other animals. Bone loss following ovariectomy in rats and nonhuman primates is much greater than in dogs. It is well recognized that fracture healing is less vigorous in old animals than in young individuals; the same is true concerning the response of the skeleton to circulating toxicants. The modeling and turnover processes present beneath the growth plates of growing animals respond to xenobiotics to a greater extent than do those in cortical or cancellous bone of older animals. In chronic studies, drug toxicity may influence (or be influenced by) the incidence and severity of spontaneous lesions occurring in the animal strain being used. For example, chronic studies with nitrofurazone show a drug-related effect that greatly increases the distribution and severity of age-related degenerative cartilage changes in rats. In the context of drug safety risk assessment, it is important to keep in mind that many test article-related effects on the growing physis represent on-target pharmacology and may not be relevant to human safety. Decisions regarding the relevance of such data depend on the proposed indication, exposure margin, and skeletal maturity of the intended population of human patients. Decreased longitudinal bone growth is a common finding in toxicity studies conducted in rapidly growing young rodents and may be secondary to decreased food intake (inanition) or occur as a consequence of test article activity. There are two main categories of disruption to endochondral ossification: physeal dysplasia and physeal dystrophy. While physeal dysplasia and physeal dystrophy reflect very different toxicologic mechanisms, both lead to functional impairment of the physis and decreased bone production and impaired growth. Hypertrophic pulmonary osteopathy (in this case, from a dog) results in extensive new bone formation beneath the periosteum in long bones. Krook, College of Veterinary Medicine, Cornell University; reproduced from Fundamentals of Toxicologic Pathology, second ed. Any substance capable of interrupting the transition from hypertrophic cartilage to the calcified cartilage spicules of primary spongiosa, whether by inhibition of angiogenesis, reduced vascular penetration, chondrocyte cell death, or a combination of these processes, can cause physeal dysplasia. Distal femoral growth plates are sometimes more severely affected than proximal tibial plates, as depicted here (C). Transverse fractures through the widened (and weak) dysplastic physis can occur through any cartilage zone. When exposure to these inhibitors ceases, these findings usually resolve to a remarkable degree in short order, although some slight residual chondrocyte column disorganization may persist. The ultimate manifestation of decreased physeal thickness is physeal closure, which occurs prematurely if the condition develops is manifested in young animals. Trabeculae of bisphosphonate-treated bone may have slightly undulating surfaces associated with these enlarged osteoclasts, which may reflect the inability of the osteoclast to produce a resorption cavity of normal (more deeply scalloped) size. Cathepsin K inhibitors produce direct antiresorptive effects on osteoclast function. Cathepsin K, an intracellular enzyme that digests type I collagen, is highly expressed in osteoclasts as a means of degrading demineralized bone matrix. Bone resorption is reduced to a similar degree as with bisphosphonates, but bone formation is enhanced because osteoclast crosstalk to osteoblasts remains intact. Retained primary spongiosa and increased trabecular bone are morphologic features of bones from cathepsin-K deficient mice. Conversely, Wnt inhibition decreases ossification, resulting in reduced trabecular bone formation in the metaphysis of growing animals. Misplaced bone removal results in trabecular splitting and a paradoxical increase in trabecular connectivity in the face of decreased trabecular bone volume by this ultimately catabolic process. Decreased numbers of osteoclasts subjacent to the zone of hypertrophic chondrocytes impair the normal elimination of extraneous calcified cartilage spicules. Primary and secondary spongiosa trabeculae numbers are increased, leading to a markedly increased trabecular bone volume in the metaphysis. Bisphosphonates are rapidly incorporated into calcified cartilage and mineralized bone, rendering these extracellular hard tissues resistant to acid digestion by osteoclasts. Exposure to bisphosphonates causes characteristic changes to osteoclasts and erosion surfaces. Osteoclasts are Decreased Osteoblast Formation/Function At high doses, first generation bisphosphonate compounds, including etidronate and clodronate, impair osteoblast function as well as osteoclast function, resulting in extremely low or effectively no bone turnover. Osteoid area and volume are augmented (increased osteoid), indicating a mineralization defect. Administration of such agents to young but skeletally mature dogs has been demonstrated to alter the structural integrity of the ribs and vertebral processes, leading to multiple spontaneous fractures. Such increases may divert these stem cells toward adipocyte differentiation and away from osteoblast differentiation. Increased adiposity of bone marrow with reduced bone formation occurs as a result of this shift. The resulting decreases in trabecular and cortical bone volume in the mouse can be dramatic. Many osteoclasts are located in lacunae on trabecular surfaces (arrows) which are normal locations, but other osteoclasts (arrowhead) are present in lacunae within the trabecular bone. As noted above, many inhibitors of chondrocyte proliferation, such as glucocorticoids, also inhibit osteoblast function. If these inhibitors have a relatively short half-life compared to the dosing interval. With respect to homeostatic bone remodeling, the decreased bone formation leads to a net bone loss since the steady state of bone resorption followed by equivalent bone formation is no longer in balance. Increased cortical porosity and decreased trabecular bone volume are typical findings with bone loss due to glucocorticoids, which can progress to osteoporosis if exposure is chronic. These effects result from the ability of steroids of this class to impair osteoblast activity. Treatment with Wnt inhibitors in animals with actively growing bones leads to a decrease in ossification beneath the growth plate, with a reduction in bony trabeculae within the metaphysis. Aberrant Bone Production Anticancer pharmaceuticals often have profound effects on bone marrow, with rapid production of woven bone appearing in the bone marrow space secondary to primary deleterious effects on hematopoietic progenitor cells. This new bone is typically noted in the middle of a segment of sternum, or in the deeper metaphysis. The mechanism for this response is thought to be stimulation from local growth factors upregulated in response to myelotoxicity. A well-known cause of abnormal bone formation is lathyrism in rats fed sweet pea (Lathyrus odoratus) or treated with its toxic agent, beta-aminopriopionitrile. This toxin inhibits lysyl oxidase, the enzyme responsible for the hydroxylation of lysine, the normal precursor to crosslinking of elastin and type I collagen fibrils. Reduced fibril crosslinking in connective tissue leads to an abnormal bone matrix and decreased bone growth and strength. Copper is a cofactor for lysyl oxidase, so copper deficiency can also depress lysyl oxidase activity. Pronounced periosteal bone proliferation (increased bone) at the tendon insertion sites of the adductus longus and pectineus on the femur is a classic finding. Systemic administration of small molecular weight peptidoglycans (typically of bacterial origin for animal arthritis models) can induce acute polyarthritis by a nonimmune-mediated process, an inflammatory response thought to be mediated by mast cell degranulation. Development of immune-mediated arthritis with exposure to peptidoglycans or other antigens requires that the antigenic material be deposited in the joint and the development of delayed hypersensitivity. One such protein, albeit incompletely characterized, has been designated "arthritogenic factor" because of its ability to sustain proliferative synovitis when instilled into the joint cavity. The joint lesion that develops following a single injection of arthritogenic factor persists for at least 4 weeks and does not require complement. The chronicity of antigen-induced arthritis depends on the persistence of a sufficient amount of antigen in the affected joint. The electronic charge of the antigen also appears to determine the development of arthritis because antigen penetration into cartilage matrix depends on both molecular weight and charge. Immune complexes trapped in collagenous tissues within joints provoke an inflammatory response; the pathologic role of sequestered antigen in maintenance of the chronic inflammatory response lies in longlasting leakage of antigen into surrounding tissue. Alternatively, complement that has leaked into the joint cavity across the inflamed synovium is activated by immune complexes. Systemically delivered therapeutic agents that are immune-stimulant compounds have been known to cause destructive arthritis in conventional studies in laboratory animals. Immune stimulants have been designed for therapy of certain systemic diseases, and adjuvants are crafted for a similar purpose with vaccines, to increase the immune response. Such compounds may cause lymphoid hyperplasia with prominent increases in B cells in lymph nodes and other lymphoid organs as well as less prominent T-cell increases in lymph nodes and spleen. Approximately 10À14 days after introduction of the adjuvant (depending on the adjuvant and antigen), rats develop swollen paws with skin discoloration. Articular cartilage was intact and appeared normal in many joints with severe peri-articular reactions although matrix loss can be substantial in apparently normal cartilage in extensively inflamed joints. Other systemically delivered compounds cause joint swelling but with a more fibroplastic response. These include bleomycin, in which the fibroplasia is considered to develop because of activation of macrophages. Rats treated with 6-sulfanilamidoindazole develop an acute exuberant synovitis and periarthritis with arteritis that can resolve in a few weeks. Loss of articular cartilage may result since normal matrix removal continues in the face of decreased synthesis. Lysosomal enzymes (collagenase, cathepsins, elastase, and arylsulfatase) are present in inflammatory, synovial, bone, and cartilage cells.
The probes must be of different sizes anxiety university california cheap buspar 5 mg with amex, and their products must be distinguishable by electrophoresis anxiety 4 hereford bull purchase buspar 10 mg amex. The protein encoded by this gene is a member of the type 3 G proteincoupled receptor family and may mediate the cellular effects of retinoic acid on the G protein signal transduction cascade anxiety tattoos buy generic buspar online. Peripheral administration of pancreatic polypeptide inhibits gastric emptying and decreases food intake in humans anxiety 60 mg cymbalta 90 mg prozac cheap buspar 5 mg buy. A number of highly plausible schizophrenia candidate genes are located in these two regions anxiety 1-10 rating scale order cheap buspar. There was another study performing cytogenomic array within 15,749 cases and 4,519 controls. These results were replicated in another Chinese sample of 399 patients with hip oF and 400 controls. Finally, two regions were successfully replicated, which are both located at chromosome 21. However, this signal was indicated to be predisposed to higher risk of fracture only in the population of European origin. Extensive normal copy number variation of a beta-defensin antimicrobial-gene cluster. Human genome sequence variation and the influence of gene history, mutation and recombination. Comprehensive characterization of human genome variation by high coverage wholegenome sequencing of forty four Caucasians. Relative impact of nucleotide and copy number variation on gene expression phenotypes. Recent duplication, domain accretion and the dynamic mutation of the human genome. Copy number variants and genetic traits: closer to the resolution of phenotypic to genotypic variability. The mechanism of expansion and the volatility it created in three pheromone gene clusters in the mouse (Mus musculus) genome. Replication stress induces genome-wide copy number changes in human cells that resemble polymorphic and pathogenic variants. High-resolution global profiling of genomic alterations with long oligonucleotide microarray. Novel approach to quantitative polymerase chain reaction using real-time detection: application to the detection of gene amplification in breast cancer. Detection of exon deletions and duplications of the mismatch repair genes in hereditary nonpolyposis colorectal cancer families using multiplex polymerase chain reaction of short fluorescent fragments. Matrix-based comparative genomic hybridization: biochips to screen for genomic imbalances. Methods to detect and analyze copy number variations at the genome-wide and locus-specific levels. Emerging themes and new challenges in defining the role of structural variation in human disease. Detection of common copy number variation with application to population clustering from next generation sequencing data. Genomic variation and gene conversion in spinal muscular atrophy: implications for disease process and clinical phenotype. Molecular definition of 22q11 deletions in 151 velo-cardio-facial syndrome patients. Six new loci associated with body mass index highlight a neuronal influence on body weight regulation. Genome-wide association study suggested copy number variation may be associated with body mass index in the Chinese population. Novel common copy number variation for early onset extreme obesity on chromosome 11q11 identified by a genome-wide analysis. Copy number variations at the Prader-Willi Syndrome region on chromosome 15 and associations with obesity in whites. Deletion 17q12 is a recurrent copy number variant that confers high risk of autism and schizophrenia. The contribution of de novo and rare inherited copy number changes to congenital heart disease in an unselected sample of children with conotruncal defects or hypoplastic left heart disease. Association analyses of 249,796 individuals reveal 18 new loci associated with body mass index. Neurotractin/kilon promotes neurite outgrowth and is expressed on reactive astrocytes after entorhinal cortex lesion. A role for pancreatic polypeptide in the regulation of gastric emptying and short-term metabolic control. Connexin 50 gene on human chromosome 1q21 is associated with schizophrenia in matched case control and family-based studies. Discovery and statistical genotyping of copy-number variation from whole-exome sequencing depth. Whole genome scanning identifies genotypes associated with recurrence and metastasis in prostate tumors. Integrated analysis of homozygous deletions, focal amplifications, and sequence alterations in breast and colorectal cancers. Genetic basis for congenital heart defects: current knowledge: a scientific statement from the American Heart Association Congenital Cardiac Defects 124. Committee, Council on Cardiovascular Disease in the Young: endorsed by the American Academy of Pediatrics. High frequency of submicroscopic genomic aberrations detected by tiling path array comparative genome hybridisation in patients with isolated congenital heart disease. An integration of genomewide association study and gene expression profiling to prioritize the discovery of novel susceptibility loci for osteoporosis-related traits. A validation of the first genome-wide association study of calcaneus ultrasound parameters in the European Male Ageing Study. A genome-wide copy number association study of osteoporotic fractures points to the 6p25. While the advantages of embracing genomic technologies to advance our understanding of gene function and association to disease may seem obvious, many scientific disciplines have lagged behind in adopting genomic approaches to further their scientific studies. A PubMed query using the keyword "genomics" returns 150,000 entries; "bone genomics" contributes less than 1% in contrast to "cancer genomics" which is the most populous category representing >17% of this total collection. Of 2100 publications associated with the keyword "bone genomics" >50% (1200) of them were published in the past 5 years, suggesting that the bone biology field is making rapid progress in embracing the new genomic technologies. However, more specialized searches, such as "osteoblast genomics" or "chondrocyte genomics," narrow the search to 235 and 170 publications, respectively, highlighting the need for the field of skeletal biology to more aggressively adopt genomic methods and technologies. Bioinformatic tools have helped predict novel genes, alternative isoforms, and protein domains, but have provided little guidance concerning the putative function of novel transcripts. In addition to defining protein domains that may be associated with known cellular functions, quantifying transcript expression across diverse cell types is critical for understanding human biology and this task is far from being exhausted. Until recently, production of such data was prohibitively expensive and experimentally laborious. Splicing arrays with probes across exonexon junctions enabled researchers to analyze predefined splicing events but could not be used to identify previously uncharacterized events. Therefore, a better understanding of the transcriptome will facilitate the extrapolation of genotypes to phenotypes and enhance our ability to discover biomarkers that can accurately predict emerging phenotypes as a function of disease progression. While this process is limited to studying the expression of validated transcripts for defined isoforms, it does however provide a glimpse of the repertoire of "genes" that are transcribed as a function of cell type, or biological process, and genome profiling in this field has led to some intriguing discoveries. However, skeletal biology is far behind in embracing the potentials of this technology compared to other fields, such as cancer biology. Comprehensive analysis of gene expression patterns and regulatory networks involved in skeletal development and remodeling is a prerequisite to completely understand physiological bone structure, function, and homeostasis. It also has a crucial role in the development of appropriate therapeutic strategies for various diseases affecting the skeleton. While many profiling reports have focused on in vitro models of cell differentiation, mineralization, or signaling perturbations, here we will primarily focus on literature that describes profiling of homogenous cell samples, tissue microdissection or organ isolation from live animals, or human samples to provide a snapshot of genomic profiling that may more faithfully adhere to in vivo bone physiology. Osteoblasts are responsible for the synthesis, deposition, and mineralization of extracellular matrix. Osteocytes are osteoblasts that have become embedded within the extracellular matrix, residing in cavities termed lacunae. Osteoclasts are large multinucleated cells that reside on bone surfaces and function primarily to resorb bone through direct chemical and enzymatic reactions. Studies of osteoblastic cells isolated from trabecular bone, embryonic calvaria, and osteosarcoma have established a set of molecular makers that help scientists distinguish the osteoblast phenotype. These markers involve the synthesis of type I collagen, the expression of alkaline phosphatase (AlP), the secretion of osteocalcin, and the production of mineralized matrix. Over the years, much attention has been given to commitment of osteoprogenitor cells, lineage progression, and differentiation into osteoblasts, processes that have been studied in various cell lines4346 and primary cultures derived from humans and rodents. However, the gene expression profiles obtained from in vitro osteogenic differentiation experiments represent only an approximation of changes occurring in vivo, which necessitates their validation in appropriate in vivo models. They are the most abundant cellular component of mature mammalian mineralized bones and constitute 1. Down: Comp, Mmp8, Ctnnb1, Wnt16, Bmp5 and Hoxa5, Hoxa7, Hoxa10, Hoxc5, Sox6, Gata1, Gata3, Cited4. Down: Hoxa1, Hoxa4, Hoxa5, Hoxa7, Hoxa10, and Hoxa13 Common to mandible and calvaria, but not ulna: Fos, Maf, Myo6, Smad3, Gata6, Ankrd1, Gli1, Nfatc4, Sox2, Gli2, Tcf7, Bmpr2, Irx2, Irx4, Klf15, Klf9, Dlx3. Down: Btc, Senp6, Crrc34, Barhl2, Tnfp, Phf20l1, Kirrel3, Crisp1, Npy2r, Krt82, Ptgs2, Pax7a, Tnf236, Crlf1 References Paic et al. Skull bone-derived osteoblasts versus limb derived osteoblasts: 249 genes Rawlinson et al. Common to mandible and ulna, but not calvaria: 873 Old versus young subjects: 446 up; 232 down 22 > 2-fold up 15 < 2-fold down kingsmill et al. Matrix formation (Up): Alpl, Bglap, Col1a2, Cthrc1, Fn1, Ibsp, Lox, Sparc, Vcan, Bgn, Fgf14, Pdgfa, Pdgfc, Pdgfrl, Pgf, Wisp1, Pthr1, Sp7. Matrix formation (Down): Efemp1, Mmp8, Prelp, Serpinb2, Spon1, Egf, Fgf1, Fgf7, Fgf23, Fgl2, Hgf, Sost, Sfrp4, Chrdl1, Grem1 (Single dose loading) Up: Wnt5a, Aspn, Igf1, Emilin2, Ccl12, Adamts4, Adamts1, Serpina3c, Stc1. Ctrl) Up: Spon1, Alox5, Baat, Sult4a1, Lp1, Mmp8, Cyp3a9, A2m, Alp1, Crabp2, Slc26a1, Cdkn2b. Down: Ibsp, Spp1, Pim1, Satb2, Akp2, Mmp9, Snf1lk, Col9a1, Runx2, Agc1, Matn4, Scgf, Dlx5, Cfh, Gtpbp2, Tubb5, Hck, Tce1, Ppp2r5d, Ndufb10, Fdps References Mantila Roosa et al. A214V/+ and lrp5+/+: 28 genes the number of probe sets changed in response to Sost antibody in any cell type or time point ranged from 0 to 514 In osteoblasts, 1650 genes clustered into 7 modules. Down: Supt5h, Ecgf1, Ifit2, Fth1, Sash1, Mrpl30, Mthfr, G1p3, Napa, Asah1, Ecm1 Farber et al. Down: Bmp2, Ramp3, Runx2, Alpl, Csf1, Ptn, Ibsp, Prpf19, Col10a1, Ngef, Ccnb2, Cdc2, Pbk. Other important differentially expressed genes: Rpl13a, Cdkn1a, Cel2, Cxcl2, Csfl1, Tnfrsf19, Arpc1a, Arpc1b, Ppp1r12a, Actn1, Thoa, Arpc5, Sfrp2, Ccnd2, Csnk2a2, Ppp2ca, Ctbp2, Tcf7, Prickl1, Rhoa, Fzd5 Up: Trem2, Anxa2, Scarb2, Ccl2, Ccl3, Cd14, St14, Ccr1, Adam9, Ptk9, Sgk1, Ctsb, Fst, Lgals1, Spp1, Sparc, Il10, Marco, Cd14, Ppp2cb, Mgl1, Rank, Shox2, Pdlim4, Hdac4, Taz. Down: Mnda, Pstp1p1, Adora2a, Lbr, Dok4, Nipbl, Nrg2, Bre, Mal, Ship1, Adora24, Irak2 Up: Dkk1, Sost, C10rf61, Sec61b, Wil1, Pcgf1, Mepe, Slc13a4. Down: Acsl3, Nips-Nap3b, Abca8, Dleu2, Ktn1, Cops4, Lrp12, Ppp3cb (All 3 tissues) Up: Sost, Dkk1, Pggf1, Ppme1. Osteoblasts and osteoclasts reside on bone surfaces, where the osteoblasts secrete extracellular matrix and contribute to osteoid formation. Osteoclasts are multinucleated cells that resorb bone through enzymatic digestion. Osteoblasts entrapped in the bone matrix, morph into osteocytes, which communicate with each other and the bone surface through cytoplasmic projections. Osteocytes are thought to be mechanosensors, meaning that they can perceive changes in tissue stress to ultimately remodel bone mass, and may coordinate the remodeling process carried out by both osteoblasts and osteoclasts. While the question of how osteocytes differ from osteoblasts at the gene expression level has been an important one, the technical limitations of in vitro assays, the lack of appropriate cell surface markers, or the inability to purify large quantities of homogenous population of cells have prevented a global analysis of their entire transcriptome. It was not until the generation of transgenic mice carrying fluorescent markers expressed by osteoblast lineage directed promoters that relatively homogeneous populations of osteoblasts and osteocytes were isolated and the gene expression profiles of these cells were examined. Prior to cell purification, the authors evaluated cell-type specificity through histological evaluation of reporter expression in calvaria of double transgenic neonatal mice. To determine the comprehensive gene expression pattern behind the process of osteoblast-to-osteocyte transformation they also compared gene expression profiles of fluorescencesorted, (Col2. Of the 3444 genes found, to be expressed in all 3 populations, 385 genes were differentially expressed between Col2. One category of genes (87 genes; 66 low/21 high in osteocytes) found to be differentially expressed between osteoblasts and osteocytes included genes encoding bone abundant extracellular proteins typically secreted by osteoblasts. Other noncollagenous structural components normally present in the extracellular matrix of bone and cartilage were also dramatically reduced in osteocytes; these included fibulins (Fbln1, Fbln2), thrombospondins (Thbs2, Thbs3), spondin 1 (Spon1), cartilage oligomeric matrix protein (Comp), proline arginine-rich and leucinerich repeat protein (Prelp), and matrilin 1 (Matn1). Several other growth factors, such as glial cell line-derived neurotrophic factor (Gdnf), growth differentiation factor 10 (Gdf10), and pleiotrophin (Ptn) had lower levels in osteocytes. While generally Sost has been described as an "osteocyte-specific negative regulator of bone formation" and is accepted as a marker of osteocyte function and activity, the authors of the osteoblast/osteocyte comparison did not find Sost among the transcripts significantly upregulated in osteocytes. Significant transcriptional differences were also observed between osteoblasts and osteocytes among 76 genes encoding plasma membrane associated proteins (52 genes twofold in osteocytes), including Phex, Ank, Gnas, Enpp1, as well as Pdpn (podoplanin), which is known as the earliest osteocyte-selective protein expressed during osteoblastosteocyte differentiation. Several members of the Notch signaling were also upregulated in osteocytes, including Notch1, Notch3, and Dlk1. Among known transcription factors, 31 were differentially expressed, 23 of which were twofold enriched in osteocytes. The group of genes with high expression in osteoblasts included Zic1, Gsc, Runx1t1, Pax1, and Scx a transcription factor with a basic helix-loop-helix motif known to function in tendons56 and developing cartilage.
Variants in the 3 or 5 invariant splice sites most commonly result in exon skipping anxiety jar buy generic buspar online, although they can also lead to activation of a cryptic splice site or intron retention anxiety symptoms one side purchase 10 mg buspar fast delivery. Clearly anxiety pill 027 purchase buspar 10 mg free shipping, splicing variants have significant impact on the protein and usually on the phenotype anxiety symptoms pdf buspar 5 mg purchase with visa. Indels are the insertion or deletion of one or several nucleotides within a sequence anxiety 36 weeks pregnant discount buspar 10 mg buy online. When the number of nucleotides is not a multiple of three, a frameshift occurs, the downstream amino acid sequence changes, and very often a new stop codon occurs downstream, truncating the protein. Thus, out-of-frame indels usually have a greater impact on protein structure and function than in-frame indels, and often impact the phenotype. Repeat expansions occur in areas within the genome that contain trinucleotide repeating sequences; an increase, or expansion, in the number of these repeating sequences can occur in both coding and noncoding regions. This type of insertion variant is known to be associated with a limited number of disease-causing genes, but constitutes an important category of variant and disease mechanism. Changes in the methylation pattern at these loci may disrupt the imprinting pattern and lead to imprinting disorders. Balanced structural variants, including chromosomal translocations and inversions, represent another type of large-scale variant. Another large-scale variant is referred to as copy neutral loss of heterozygosity (cn-loH). Identity by descent may be due to distant relatedness between two individuals or consanguinity indicating recent relatedness. Thus, the strength of amplification signal reflects the amount of genomic target (inferred as copy number). Due to the limited resolution of cytogenetic approaches, the balanced genomic variants are believed to be underdetected. It should be noted that many apparently balanced rearrangements ascertained by cytogenetic approaches are later found to harbor cryptic imbalances when high resolution microarraybased genomic profiling techniques are used. Many challenges lie ahead, particularly in the area of data interpretation (informatics), which often involves database searching, segregation analysis, bioinformatic prediction, and functional demonstration. The nature of the variant dictates what method should be used for genetic testing. Conventional approaches to genetic testing in the clinical setting involve interrogating one gene or one variant at a time in one patient. The molecular diagnostic laboratory may use Sanger sequencing to test for small-scale variants, and whole gene or gene-panel based tests are currently the most commonly used approaches in genetic testing. GeneTiC TesTinG and CounselinG unknown genetic conditions, and microarray-based genomic profiling techniques have been recommended as the first tier genetic test in many clinical scenarios. Although there has always been a lag between the research discovery of causative genes and the availability of commercial diagnostic testing,28 genetic testing is available for a large number of skeletal diseases, including metabolic bone disorders and skeletal dysplasias. Genetic testing for metabolic disorders of bone can be useful in the clinical setting for diagnostic purposes. There are a number of genetic causes of metabolic bone disease for which there is genetic testing available, including familial hypophosphatemic rickets, autosomal dominant hypophosphatemic rickets, two forms of autosomal recessive hypophosphatemic rickets, X-linked recessive hypophosphatemic rickets, and hereditary hypophosphatemic rickets with hypercalciuria. Finally, genetic testing is available for hypophosphatasia, due to variants in AlPl, the gene encoding the alkaline phosphatase, tissue nonspecific isozyme. For many of these conditions, the diagnosis is made clinically and can be confirmed by genetic testing. Genetic testing for skeletal dysplasias can be used to confirm a clinical diagnosis, as well as for cases where a clinical diagnosis is not immediately clear but several different disorders are under consideration. All patients with a suspected skeletal dysplasia should have a thorough physical exam documenting any dysmorphic features, as well as a full radiographic skeletal survey in cases where the diagnosis is not suggested by history and physical exam alone. Prior to ordering any genetic test, the provider needs to determine if the desired test is clinically available. The most common clinical situation where genetic testing is ordered is when the genetic testing is required to make a diagnosis or to confirm the diagnosis. Genetic testing is especially useful if a course of treatment is available and dependent on the diagnosis. In these cases, genetic testing will provide a clear benefit for the care of the affected patient. A clear example of this is osteogenesis imperfecta, in which bisphosphonates may be beneficial. This involves requesting one genetic test at a time and moving on to other genetic testing only if the initial tests are negative. This staged testing approach is also useful for disorders with genetic heterogeneity. Testing can begin with the most commonly affected gene and then proceed to subsequent genes only if the first test is negative. However, in cases where diagnosis will affect treatment and delaying therapy is not optimal, ordering multiple tests at once or gene panels may benefit the patient. Reproductive counseling is another reason that genetic testing may be useful in a clinical setting. It is not uncommon for skeletal disorders to be diagnosed clinically based on family history, physical exam, and radiographic evidence. While the diagnosis may not be in question, knowledge of the specific variant causing the phenotype may be useful for future reproductive decisions. In the case of recessive disorders, carrier testing is appropriate for adult family members but should not be performed on minor children until they reach maturity and can consent to their own testing. In cases where genetic testing is needed for initial diagnosis or to confirm a clinical diagnosis, a full discussion of the risks and benefits should be undertaken with the patient (or parents if the patient is a minor). Discussion of these issues can help alleviate many misunderstandings and anxieties about genetic testing. Available genetic tests may only detect variants in a small number of clinically diagnosed patients depending on the disorder. Patients and families may consider a negative test result "the final answer" and need to understand that a negative genetic test does not always exclude a given clinical diagnosis. The cost of genetic testing can affect the ability to order genetic tests in different settings. Genetic tests can be among the most expensive laboratory-based tests available, ranging from hundreds to tens of thousands of dollars. Third-party payers are becoming increasingly reluctant to pay for the costs of genetic testing, particularly in the absence of documented clinical necessity. However, families with high-deductible plans should be warned that the cost of one genetic test may be greater than the entire deductible, requiring a single large payment. Carrier testing may not be covered by insurance companies, except when done for prenatal testing purposes. This can effectively put testing for some disorders out of the reach of patients and providers. These increasing costs place a responsibility on providers to ensure that genetic tests are only ordered under appropriate circumstances where there is a clear benefit for the patient. The increasing availability of genetic testing for all types of skeletal disorders can be of great benefit to patients and providers. Genetic testing can be useful for diagnosis and subsequent treatment, as well as for reproductive counseling for patients and families. However, as with all medical testing, genetic testing has risks and benefits and may not be appropriate for every clinical situation. As much of the current research in genetics and genomics is focused on the study of complex trait versus single-gene disorders, the possibility that there will soon be numerous additional clinical tests available is not out of the question. Given the advances in genetic and genomic medicine, subspecialists, as well as primary care physicians have been increasingly being called upon to utilize genetic information, including risk assessment and testing in their practice38,39 and help to determine which patients may be appropriate for genetic testing and counseling. The diagnosis of a genetic disorder is likely to bring up numerous questions for patients and their families. Families must grapple with the many facets of the diagnosis, including the natural history, prognosis, and treatment options. Genetics professionals can help individuals to navigate these issues by providing the factual information required, as well as engaging patients and their families in a discussion of the potential psychological, social, and health impacts of the disorder. While primary care providers and subspecialists offer education concerning the clinical features, treatment, and prognosis of bone disease, genetics professionals are 1. GeneTiC TesTinG and CounselinG often needed to ensure that patients and their families are receiving comprehensive information regarding the testing, therapeutic, and reproductive options available to them, as well as psychosocial support. As such, it becomes incumbent upon primary care clinicians, genetics professionals, and other clinical specialists to partner to provide optimal genetics care, including counseling, to their patients. The term "genetic counseling" was coined in 1947 by Sheldon Reed, who subsequently authored the book Counseling in Medical Genetics. Therefore, in the early years of genetic counseling, families could only be provided with medical information and predictions of recurrence risk. Genetic counseling is carried out by genetics professionals, generally clinical geneticists and genetic counselors. Alternatively, physicians can complete a 45 year training residency programs that combines Clinical Genetics and Genomics with Internal Medicine, Maternal Fetal Medicine, Reproductive endocrinology and Infertility, or Pediatrics. Graduates of these genetic counseling programs are trained to provide services to patients and their families, and receive instruction in human, medical, and clinical genetics, as well as counseling theory and techniques. The training of all genetics professionals also includes exploration of the relevant social, ethical, and legal issues inherent in the field. As noted earlier, practitioners of genetic counseling or "genetics professionals" include genetic counselors, clinical geneticists, genetic nurses, and other clinicians with specialized training in the field. While genetics providers have traditionally worked in academic medical institutions, genetics professionals also provide services within the primary care, diagnostic laboratory, and public health sectors, as well as in specialized areas, including assisted reproductive technologies, cardiology, and oncology. As such, genetics providers serve patients at all stages from preconception through pregnancy to pediatrics through adulthood. However, this approach has been questioned, both in terms of its definition, as well as its role in the genetic counseling session. However, there may be situations when this may not be appropriate, such as discussing a particular diagnosis that has a well-established treatment strategy. A patient may be referred to the genetics professional with a diagnosis that needs to be confirmed, or the patient may be referred to a clinical geneticist to make a genetic diagnosis. Regardless of the setting, the components of the genetic counseling session are similar. This allows the genetics provider to dispel any initial misconceptions regarding the purpose and potential outcomes of the genetic counseling session including genetic testing. The "pedigree process" also allows one to correct any misapprehensions the patient may have about the degree of heritability in a family for any given diagnosis. Families may have long-held beliefs regarding the causes of certain disorders within the family. For example, family members may falsely attribute birth trauma as the cause of a disorder, or believe that a certain disorder affects only males since all of the females in their family are unaffected. Therefore, it is important that the genetics provider initially determines how the family has come to such beliefs to address any misconceptions in a sensitive yet effective manner. The duty to warn relatives with respect to their genetic risk is an ethical issue that often needs to be addressed during this portion of the genetic counseling session. This duty may bring up a multitude of concerns, including patient confidentiality and the autonomy of relatives in being able to choose not to know of their risk status. However, as the discussion of the family history includes the possible identification of at risk family members not present in the session, the potential impact the disorder may have on the medical management of those family members cannot be ignored. While the focus of the counseling session should remain on the patient and the family members present, genetic counseling needs to include guidance as to why and how to discuss the diagnosis and potential impact with other family members, and to guide them to appropriate resources. As such, patients must be counseled as to the real and potential risks, benefits, and alternatives to testing, and also be given the assistance needed to make an informed decision regarding their testing options. It is worth noting that patients may actually put more weight on their perceived risk and 1. Pretest genetic counseling also allows for a discussion of the potential for unclear or unexpected findings, such as variants of unknown significance, incidental/ secondary findings, and nonpaternity. Genetic testing in the setting of childhood and adolescence is an issue that continues to be debated in the genetics field. Multiple professional societies have taken the stance that testing of minors should only be done when the disorder in question has a childhood onset and effective treatment or surveillance in childhood is available. The potential benefits, such as elimination or reduction of uncertainty, or the initiation or elimination of surveillance techniques, must be weighed against the potential harms, such as increased anxiety or effects on familial relationships. Another issue that has been highlighted with regard to genetic testing is the "duty to recontact. Patients may also need to be recontacted in the setting of previously identified variant of unknown significance being reclassified as a causal or "normal" variant. Does a genetics clinic have the appropriate infrastructure and resources required to recontact all relevant patients with new testing options How will the primary care provider keep current information on the development of new genetic tests Currently, as outlined by the American College of Medical Genetics and Genomics policy statement, the genetics provider should initiate the recontact in cases where there is an ongoing relationship with the patient, whereas in cases lacking this ongoing relationship, the primary care provider should shoulder the responsibility. Genetic testing poses unique challenges as the disclosure of genetic information, whether voluntary or involuntary, has the potential to lead to social and economical consequences. However, as noted previously, it does not speak to the possibility of genetic discrimination with regard to life, disability, or longterm care insurance. Genetics providers operate under the belief of complete disclosure; families should be given all relevant information as it pertains to the diagnosis. In addition, it may not be appropriate or feasible to disclose every piece of information regarding a disorder, genetic test, or treatment as the provision of too much information during a single session may lead to confusion and negatively 1. For example, when providing prenatal genetic counseling to a couple who already have a child with a skeletal dysplasia, a detailed explanation of prenatal testing options may be more appropriate than a lengthy discussion of the prognosis of the disorder.
However anxiety of death quality 5 mg buspar, the inclusion of the entire genome results in several challenges anxiety quick fix purchase buspar 10 mg with mastercard, not least the huge amount of data generated and the requirement for bioinformatics expertise in its analysis anxiety 411 discount buspar 5 mg buy on line. Indeed anxiety symptoms 100 order buspar 10 mg without a prescription, WeS has been the mainstay of highly successful diseasegene discovery studies over the past decade anxiety symptoms of flu 10 mg buspar purchase mastercard, including the identification of several genes responsible for skeletal dysplasias and metabolic bone disorders [e. Furthermore, the more manageable amount of sequence data makes analysis more straightforward, although this still may present challenges (see later). The principle is similar to that of WeS, but only specific genes of interest undergo capture with customdesigned probes. However, its limitations include the requirement to know in advance the potential causative genes. In these settings, ascribing pathogenicity to individual variants may be challenging and as a result, guidelines have been recommended by several international bodies suggesting a systematic approach to variant analysis. These guidelines recommend that all variant classification systems utilize the terms "pathogenic," "variant of uncertain significance (VuS)," and "not pathogenic or benign," and that evidence for pathogenicity is gathered from multiple sources. These potential considerations and processes that are typically included in the variant identification and interpretation pipeline are briefly reviewed, below. This begins with the alignment of the sequence reads to a reference genome, which should be obtained from a 1. In most instances it is reasonable to exclude all synonymous variants from further analysis. This is particularly pertinent in rare diseases in which the frequency of a variant in the population database should not exceed that within the disease population. Where possible the primary source of information supporting previous reports of variant pathogenicity should be consulted. Segregation in close family members is not evidence of causality per se, as the variant may actually be in linkage disequilibrium with the causative gene, and thus samples should be sought also from distant family members if possible. Likewise, in the research setting, prior cosegregation studies in a family, which may have identified putative chromosomal locations shared by affected individuals, may markedly narrow the focus on specific variants. Further investigation of variants for likely functional effects relies on an assessment using computational tools and/or employing in vitro and or in vivo assays. Thus, variants can be subdivided into: those that are missense, nonsense, or frameshift variants; those within exons and introns predicted to affect splice sites; and those within uTrs. One can then assume that nonsense, frameshift, and splicing mutations that lead to whole exon deletions are more likely to be pathogenic. However, the biological function of the gene must also be considered as some diseases are only caused by heterozygous missense mutations, while many genes harbor high frequencies of heterozygous null variants without any detrimental effect on health. For evaluation of nonsynonymous variants, an evaluation of the population frequency is an essential step, typically allowing the exclusion of variants which are observed with an allele frequency of >1%. Concurrently, it is important to consider the clinical evidence for the pathogenicity by evaluating whether the variant cosegregates with affected individuals in the kindred. Variants are then divided by functional type into missense, nonsense, splice site variants, or those affecting untranslated regions (uTrs), as approaches to analyses differ for each variant class. For missense and nonsense variants, a number of computational analysis tools exist to examine frequency in clinical populations, evolutionary conservation and the effect on protein structure. While these can be informative, they should not be used as the only indicator of pathogenicity as they have low specificity. Identification of evolutionary conservation or location of the variant residue within a critical structural domain of the affected protein can also provide evidence that a variant may be pathogenic. Several computational tools are available to assess these parameters including ClustalOmega that can align sequences based on homology, the Protein Data Bank that catalogs published protein structures, and Pymol that can be used to model structures downloaded from Protein Data Bank. For example, if the gene is known to regulate a signaling pathway already known to be important in the condition. Once a variant has been assessed by these bioinformatics tools and still remains a plausible candidate, functional analysis should be performed. Some care must be taken to select the appropriate experimental technique and this may depend on: the type of variant. In choosing an in vitro assay the heritability of the variant should also be considered as overexpression systems may exaggerate the effect of a variant, and therefore may not accurately reflect the effect of a heterozygous variant. Further important evidence to implicate a gene causing a disease can come from screening additional affected families and individuals for further mutations in the gene. Mosaicism refers to an individual who has developed from a single fertilized egg but has two or more populations of genetically distinct cells. In this setting, apparently unaffected parents (with one carrying the mutation limited to their gametes) may give rise to more than one affected child, suggesting possible autosomal recessive inheritance, in contrast to the underlying autosomal dominant inheritance pattern. Specifically, these tests provide far greater sensitivity to detect low levels of mosaicism. However, the most appropriate diagnostic test will depend on the clinical picture, the suspected type of mutation. The potential indications, nature, and timing of genetic testing will therefore depend on the clinical scenario. Preimplantation genetic diagnosis: For individuals at risk of a serious genetic disorder, in vitro fertilization offers the opportunity to screen embryos for the specific defect prior to establishing pregnancy. In this process, a few days after egg fertilization, a single cell is removed from the embryo for genetic analysis, which allows the detection of major chromosomal abnormalities. Screening of serial embryos allows up to two unaffected embryos to be subsequently implanted for pregnancy to develop. These samples have been used in tests that include: conventional karyotyping for the detection of aneuploidy. Currently, in the clinic it is most frequently used to screen for aneuploidy (Down, edwards, Patau, and Turner syndromes) and for fetal sex determination (important for X-linked disorders or conditions that severely affect one sex, for example, females with congenital adrenal hyperplasia). However, such genetic testing may also give rise to clinical and ethical challenges. Communicating this uncertainty to the patient (and perhaps the wider family) remains a major clinical challenge. However, it is clear that the likelihood of such ambiguous results should constitute part of informed consent prior to genetic testing. In addition, clinicians and academics both have a responsibility for accurate reporting of genetic data coupled with phenotype data to improve the reliability of disease-specific mutation databases. For example, the inadvertent identification of penetrant pathogenic mutations in hereditary cancer genes. Next-generation sequencing of common osteogenesis imperfecta-related genes in clinical practice. Whole-genome sequencing identifies eN1 as a determinant of bone density and fracture. Standards and guidelines for the interpretation of sequence variants: a joint consensus 48 49. Consensus Statement on next-generationsequencing-based diagnostic testing of hereditary phaeochromocytomas and paragangliomas. Systematic comparison of variant calling pipelines using gold standard personal exome variants. Databases of genomic variation and phenotypes: existing resources and future needs. Genetic variations that cannot be tolerated can cause susceptibility to various disease states and abnormal functions. Genetic linkage studies and targeted sequencing together with recent advances, such as genome-wide association study and next-generation sequencing approaches have provided valuable insight into normal and abnormal genetic variations (see Chapter 3). In 1942, the developmental biologist Conrad Waddington used the term "epigenetic landscape" to describe the phenotypic alterations that occur from cell to cell during development in multicellular organisms (this article from 1942 was reprinted in 2012). Unlike genetic alterations epigenetic changes are reversible, a property that is desirable for the development of drugs that can combat disease states associated with epigenetic defects. This article presents a general background of epigenetic control mechanisms and the contribution of epigenetics to human disease. They contain a globular domain and a flexible unstructured amino terminal tail (the histone tail) that protrudes from the nucleosome. Enzymes of the histone kinase family phosphorylate specific serine, threonine, or tyrosine residues, and phosphorylation can be reversed by the action of phosphatases. For example, di- or trimethylation of histone H3 at lysine 4 (H3k4me2 and H3k4me3) and monomethylation of H3k9 (H3k9me1) are marks of active transcription. The amino acid position in the histone protein sequence is written below each amino acid. TrxG proteins also exist as several multimeric protein complexes containing histone methyltransferase 1. EpigEnEtics activity that are associated with active chromatin states where they induce trimethylation of histone H3 at lysine 4 (H3k4me3). Therefore, the functions of the PcG and TrxG proteins are described as "antagonistic" to each other promoting transcription repression and activation, respectively. Histone variants and their modifications contribute to nucleosome and chromatin structure and function. CpG islands often occur in promoter regions and their methylation usually prevents gene expression. Epigenetic modifications can be passed on to daughter cells somatically after cell division. This reprogramming resets the epigenome (epigenetic marks across the entire genome) of the early embryo to render the cells totipotent (the potential to form every cell type in the body). To pass to the next generation through the germline, epigenetic marks must avoid being erased during embryonic reprogramming. However, a subset of "imprinted" genes are expressed in a monoallelic parent-of-origin dependent manner in which one parental allele is partially or completely silenced. EpigEnEtics imprinting causing two active copies of a gene or from inactivation of the nonimprinted allele causing two inactive copies. Comprehensive characterization of genome-wide epigenetic marks has been informative to reveal the status of the epigenome in various diseases, particularly cancers. Targeting the disease-associated epigenome holds promise irrespective of whether it is the cause or consequence of the disease process. Other classes of drugs under development and/or in clinical trials are those that target-specific interactions in chromatin modification protein complexes or specific epigenetic modifications. The interplay of these mechanisms in intra- and internucleosomal interactions over short and long distances generate a variety of chromatin states. The sum of these mechanisms is fundamental to the regulation of diverse cellular processes through differential transcriptional readout of the same genetic material. The importance of epigenetics is underscored by many diseases that can develop due to mutations in epigenetic regulatory proteins, misregulation of the epigenetic machinery, and aberrant placement or removal of epigenetic marks. The reversible nature of epigenetic alterations is an attractive target for therapeutics that can help reset the epigenome to the normal state. The fact that some of these epigenetic drugs have been efficacious in the treatment of cancers, such as hematological malignancies reinforces the importance of epigenetics. The recent developments in high throughput sequencing techniques have enabled epigenome profiling of various cell types in their normal or pathological states. These epigenome signatures can be valuable for disease diagnosis, prognosis, and treatment opportunities. Ongoing and future research in the field hopes to shed light on epigenetic changes from a host of inputs, such as aging, metabolic, nutritional, physiological states, environmental conditions, early and late life exposures, chemical, and immunological challenges. Histone phosphorylation: a chromatin modification involved in diverse nuclear events. Peptidylarginine deiminases in citrullination, gene regulation, health and pathogenesis. A maternal-zygotic effect gene, Zfp57, maintains both maternal and paternal imprints. Trim28 is required for epigenetic stability during mouse oocyte to embryo transition. Distinct H3F3A and H3F3B driver mutations define chondroblastoma and giant cell tumor of bone. Histone H3k36 mutations promote sarcomagenesis through altered histone methylation landscape. Even if such loci were established, linkage approaches lacked sufficient resolution to pinpoint the responsible genetic variants underneath the linkage peaks. These features include: better quality control of genotypes and cohorts, including identification of cryptic relatedness among the cohort, identification and control of population stratification, and generally more stringent exclusion of poorly performing samples or markers. Considerably more stringent statistical thresholds are used for claiming identification of positive findings, with genomewide significance set at P < 5 × 10-8. However, Risch and Merikangas suggested that testing a million alleles for association in cohorts of achievable size (<1000 individuals) would be an efficient and powerful way to detect genes with modest effects (genotypic relative risk of 1. Although this remains a stringent threshold for declaring association, most geneticists still regard significant association as proven only when replicated in an independent cohort. While this may be mathematically true, a high proportion of findings at lower significance values prove to be false positives, and thus candidate gene studies achieving "experiment-wise" significance should be interpreted with some caution, if genome-wide significance is not achieved. At the time of their publication, the study design proposed by Risch and Merikangas was theoretical, as the full extent of variability across the genome was unknown and an efficient means of capturing that variability was technically impossible. Major technological advances in fields as diverse as chemistry, optics, and computational processing lead to high-throughput array genotyping; and large collaborative genetic programs, such as the International HapMap and Human Genome Project characterized the extent of variability in the genome. The International HapMap project was established to characterize the haplotypic structure of the genome [initially for just 4 populations (white Europeans, Chinese, Japanese, and Yoruban Nigerians) but now including 11 other global ancestry groups]. This argument assumes that common diseases are caused by common variants in the population that have arisen in a common founder to whom carriers of the disease-associated variant are distantly related, the socalled "common diseasecommon variant" hypothesis. To illustrate how this might occur, at some point in the past an ancestor developed a genetic mutation that resulted in a disease. If all current individuals with this disease have arisen from this common founder, then all 1.
Order cheap buspar on line. Joel Osteen - Keep Your Joy.
References
- Sparano JA, Gray RJ, Makower DF, et al: Adjuvant chemotherapy guided by a 21-gene expression assay in breast cancer, N Engl J Med 379:111n121, 2018.
- Wilcox WR, Khalaf AA. Nucleation of monosodium urate crystals. Ann Rheum Dis 1975; 34:332-9.
- White H, Venkatesh B. Cerebral perfusion pressure in neurotrauma: a review. Anesth Analg. 2008;107(3):979-988.
- Schotte CK, Van Den Bossche B, De Doncker D, et al. A biopsychosocial model as a guide for psychoeducation and treatment of depression. Depress Anxiety. 2006;23: 312-24.
- Moorjani GR, Michael AA, Peisjovich A, et al: Patient pain and tissue trauma during syringe procedures: a randomized controlled trial. J Rheumatol 35:1124-1129, 2008.
