
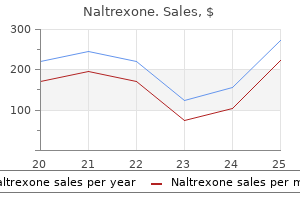
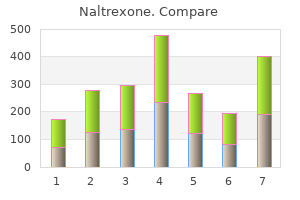
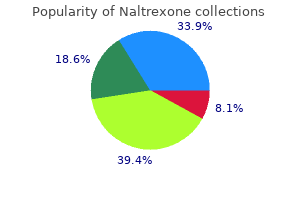
Naltrexone
| Contato
Página Inicial
Sheryl G.A. Gabram, MD, MBA
- Professor of Surgery
- Department of Surgery
- Emory University
- Director
- AVON Comprehensive Breast Center at Grady
- Winship Cancer Institute at Grady
- Atlanta, Georgia
Specific selection of the injured renal artery branch is ideal before embolization because some degree of renal infarction usually occurs symptoms uterine cancer generic naltrexone 50 mg buy on line. These include the nature of the vascular injury medicine and technology naltrexone 50 mg buy with amex, the size of the injured artery treatment tendonitis generic 50 mg naltrexone amex, and the flow dynamics of the vessel medicine universities order 50 mg naltrexone amex. Gelatin sponge has been used most commonly for embolization because of its utility medications you can give your cat buy naltrexone 50 mg with visa, absorbability, ease of use, and familiarity to operators. This allows for embolization of the selected branch of the renal artery as well as its small distal tributaries with minimal associated nontarget embolization. Alternatively, the venous outflow tract can be temporarily stopped with a balloon occlusion catheter. There is an abrupt occlusion of the right main renal artery with surrounding hematoma. Abdominal aortogram (B) demonstrates abrupt cutoff of the main renal artery with nonperfusion of the right kidney. After the occlusion was carefully traversed with a guide wire, advancement of the catheter into the renal artery (C) revealed avulsion of the artery with free extravasation of contrast material and no filling of the distal renal artery. Alternatively, direct injection of thrombogenic material can be performed via an ultrasound-guided percutaneous approach. This could be performed if the bleeding vessel cannot be accessed with a microcatheter. In a series of 14 patients, direct percutaneous embolization of traumatic pseudoaneurysms with gelatin sponge was successful in 13 patients. Angiography was performed 90 minutes after the trauma, showing complete occlusion of the left main renal artery with selective catheterization (B). Extreme care should be taken when selecting the renal artery with a diagnostic catheter so as not to cause further injury or vasospasm. In the case of a renal artery dissection, attempts should be made to cross the dissection with a wire. Once the diagnostic catheter is removed, angiography through the sheath can be performed to plan for potential stent placement. Since then, numerous series have been published describing the success of transarterial embolization (Table 75. Gelatin sponge pledgets were used for the smaller lesions, whereas steel coils were used for the larger ones. However, 90% demonstrated some degree of parenchymal infarction, including 33% of cases with greater than 50% parenchymal infarction. At that time, microcatheters were not in routine use, which could have allowed for superselective catheterization, reducing the degree of parenchymal infarction. Four of the nine patients had injury to the main renal artery, whereas the remainder had injury to the renal parenchyma. Of the nine patients, seven underwent complete embolization of the main renal artery and two patients underwent selective embolization. The operators obtained both clinical and technical success in all cases with no patients requiring further intervention, including surgery. At the time of long-term follow-up (mean 32 months) there were no cases of renal failure. In another retrospective series of 13 patients,42 both technical and clinical successes were achieved in 100% of the patients. Hagiwara and colleagues43 performed the only prospective study evaluating angiography with embolization in this setting. One of the 21 patients Contraindications There are no absolute contraindications to renal embolization. In patients with single kidneys, attempts should be made to perform selective embolization to avoid complete renal failure. Nephropathy secondary to contrast use may be challenging to diagnose because concurrent renal injury is also present. Perhaps of greatest concern is nontarget embolization with material inadvertently distributing to alternate areas of the kidney or to other locations downstream in the aorta. Furthermore, the actual placement of coils may displace the catheter and result in a more central occlusion, producing a larger area of infarction. Postembolic hypertension is a rare complication that has been observed after surgical treatment. However, there have been no reported cases in the literature following catheter-directed therapy. Postembolic infection has not been a factor, but application of good sterile technique continues to be essential in any embolization procedure. This requires aggressive hydration and pain control with narcotics via patient-controlled analgesia if not contraindicated. Postembolization infarction syndrome manifests as pain, leukocytosis, and transient low-grade fever. Treatment is supportive, consisting of hydration, antipyretics, antiemetics, and narcotics. In the case of complete renal embolization, surgical nephrectomy can be performed when the patient stabilizes. Deformity of the kidney by a large intrarenal hematoma can occur with stretching of vessels, which in turn may prevent subselection and necessitate a more proximal embolization with more tissue loss caused by infarction. Overall, the authors were able to avoid exploratory laparotomy in greater than 90% of the patients. Renal artery stent placement in the setting of renal artery injury in trauma was initially described in 1995 by Whigham. Additionally, endovascular restoration of the renal artery in renovascular trauma may be considered more. In severe cases (grade V), most patients are intervened surgically, often with nephrectomy. In the remainder of cases, catheter-based therapy can play a significant role in treating hemorrhage and stabilizing patients. Its increasing use, increasing amount of evidence, and the more conservative approach taken toward kidney trauma will result in a greater role for the interventional radiologist in this setting. In the rare cases of renal artery injury, catheter-directed therapy can play a significant role if intervention can be performed in a timely manner. Renal injury and operative management in the United States: results of a population-based study. Management and hospital outcomes of blunt renal artery injuries: analysis of 517 patients from the National Trauma Data Bank. Renal and extrarenal predictors of nephrectomy from the national trauma data bank. Radiographic predictors of need for angiographic embolization after traumatic renal injury. Validation of the American Association for the Surgery of Trauma organ injury severity scale for the kidney. American Association for the Surgery of Trauma grade 4 renal injury substratification into grades 4a (low risk) and 4b (high risk). Percutaneous embolization for the management of grade 5 renal trauma in hemodynamically unstable patients: initial experience. Intermediate-term follow-up of patients treated with percutaneous embolization for grade 5 blunt renal trauma. Outcome after major renovascular injuries: a Western trauma association multicenter report. Stab wounds of the renal artery branches: angiographic diagnosis and treatment by embolization. Use of the Palmaz stent in primary treatment of renal artery intimal injury secondary to blunt trauma. Successful management of postbiopsy arteriovenous fistula with selective arterial embolization. Transcatheter embolization of biopsy-related vascular injuries in renal allografts. The role of interventional radiology in the management of blunt renal injury: a practical protocol. Traumatic intimal tear of the renal artery treated by insertion of a Palmaz stent. Treatment of posttraumatic dissection of the renal artery with endoprosthesis in a 15-year-old girl. Increasing role of angiography and segmental artery embolization in the management of renal stab wounds. Blunt renal trauma: minimally invasive management with microcatheter embolization experience in nine patients. Effects of superselective embolization for renal vascular injuries on renal parenchyma and function. Vascular injuries after percutaneous renal procedures: treatment by transcatheter embolization. Up to 60% of the population experiences at least one episode of nasal bleeding, but only 6% require medical care. Anterior epistaxis is the most common type and is located at the conjunction of the anterior septum vessels, an area known as Kiesselbach plexus. This location is readily accessible and treatable with simple measures, such as manual compression, anterior nasal packing, or cautery. Because it is located in a relatively inaccessible region, treatment is not so simple and usually requires posterior nasal packing and hospitalization. In such a situation, occlusion of the arteries supplying the posterior nasal fossa is necessary to stop the bleeding; therapy could be performed either by surgery or by endovascular embolization. Although still controversial in some institutions, embolization has gained increased acceptance and large series with excellent results have been reported. Although it may occur at any age, there is a mild peak of onset before 20 years of age and a more remarkable age-related increase after 40 years of age. In many cases, the cause of bleeding cannot be determined-so-called idiopathic epistaxis-but hypertension, heavy smoking, alcohol consumption, and hypercholesteremia have been described as associated factors. In other circumstances, there is a well-recognized etiology of nasopharyngeal bleeding that may be diagnosed by proper anamnesis, clinical examination, or imaging modalities: vascular malformations, trauma, hypervascular tumors, and systemic coagulopathy are among the most common. Other vascular malformations causing epistaxis are less common but aneurysms, arteriovenous, or venous malformations should be considered. It should be noted that packing is painful and stressful for the patient and its prolonged use may be complicated by infection, alar necrosis, aspiration, and respiratory disorders. Indeed, the main danger in head and neck embolization lies in lack of familiarity with the anatomic vascular arrangement in a given patient. The transosseous arteries at the base of the skull that communicate the extra-intracranial circulation are of utmost concern and therefore should be clearly recognized by the interventional radiologist. It ramifies into two major groups, the medial branches that supply the nasal septum and the lateral branches that supply the turbinates and the paranasal sinuses. These extra-intracranial connections, although not always seen at angiography, are always present because they correspond to the vascular embryologic remnants of the neural crest. In young individuals or patients with hypervascular nasopharyngeal lesions, a single diagnostic 4 French catheter could be used for both diagnostic angiography and embolization. The guide wires that are commonly used depend on the catheter scheduled, usually a 0. The embolic material varies depending on the etiology of the epistaxis and the angioarchitecture of a given lesion. The latter may embolize too distally with an increased risk of necrosis, cranial palsy, and undesired passage to transcranial anastomoses. Treatment strategy selection depends on many factors, such as type of bleeding, hemodynamic situation of the patient, arterial source, clinical history, as well as medical expertise and/or local preferences. In institutions where skull base endovascular therapy is commonly performed, embolization is usually the preferred treatment because of its multiple advantages. The procedure, including angiography and embolization, is performed in the angiographic suite with the nasal packing in place. Although a conscious sedation or local anesthesia may be alternative possibilities, it should be remembered that the nasal packing is uncomfortable and could lead to respiratory insufficiency or aspiration in these anxious patients. In the latter, patients need to be awake and cooperative for clinical test occlusion. Clinical care (transfusions, hemodynamic stabilization, correction of coagulation disorders, etc. If bleeding stops, the nasal packing is removed 48 hours after placement, and if there is no recurrence the patient is discharged with further medical control as an outpatient. In such a case either endoscopic surgery or transarterial embolization is currently the best way to achieve this goal. Diagnostic pretherapeutic angiography and therapeutic embolization are performed not in a random fashion but following a standardized protocol that may vary depending on the clinical suspicion of bleeding site and type of epistaxis. The standardized angiographic and embolization protocol that is employed at our institution for idiopathic epistaxis is widely used and has been previously described. The angiographic and embolization protocol and its key points are summarized in Table 76. If important nasal vascular supply is present, embolization is immediately performed; if not, some authors do not embolize but others prefer embolization anyway to decrease the risk of early recurrence of epistaxis. Idiopathic Epistaxis this is the most common type of intractable epistaxis and usually the angiographic findings are normal. It is not expected, as is the case in other severe hemorrhages (gastrointestinal, splenic, etc. Occasionally, congestion or hyperemia in the nasal mucosa may be identified due to local inflammatory changes and the presence of the nasal packing. The aforementioned protocol of angiography and embolization is recommended to achieve a successful result to stop this type of bleeding. A Hereditary Hemorrhagic Telangiectasia this entity, also known as Rendu-Osler-Weber disease, is a systemic autosomal dominant hereditary disorder involving vascular abnormalities in various organs.
Mechanical thrombolysis of thrombosed hemodialysis native fistulas with use of the Arrow-Trerotola percutaneous thrombolytic device: our preliminary experience medicine kit for babies naltrexone 50 mg order online. Procedural success and patency after percutaneous treatment of thrombosed autogenous arteriovenous dialysis fistulas treatment 11mm kidney stone buy naltrexone with amex. Percutaneous thrombolysis of thrombosed haemodialysis access grafts: comparison of three mechanical devices medicine evolution naltrexone 50 mg order free shipping. Effectiveness and safety of dialysis vascular access procedures performed by interventional nephrologists medicine rocks state park purchase naltrexone 50 mg. Ventilation-perfusion scintigraphic evaluation of pulmonary clot burden after percutaneous thrombolysis of clotted hemodialysis access grafts treatment hepatitis b buy naltrexone online now. Pulmonary embolism from pulse-spray pharmacomechanical thrombolysis of clotted hemodialysis grafts: urokinase versus heparinized saline. Backbleeding technique for treatment of arterial emboli resulting from dialysis graft thrombolysis. Upper limb ischemia after vascular access surgery: differential diagnosis and management. Arterial percutaneous angioplasty in upper limbs with vascular access devices for haemodialysis. Treatment of ischemia due to "steal" by arteriovenous fistula with distal artery ligation and revascularization. Hand ischemia in patients with hemodialysis access grafts: angiographic diagnosis and treatment. Aneurysms of hemodialysis access grafts: treatment with covered stents: a report of three cases. Prolonging the life of difficult hemodialysis access using thrombolysis, angiography and angioplasty. Efficacy of covered stent placement for central venous occlusive disease in hemodialysis patients. The impact of a community-wide vascular access program on the management of graft thromboses in a dialysis population of 495 patients. Effect of clopidogrel on early failure of arteriovenous fistulas for hemodialysis: a randomized controlled trial. Cough, hoarseness, dyspnea, stridor, and dysphagia as manifestations of laryngeal and/or pharyngeal edema are seen. Other findings also include those caused by cerebral edema, such as headache, confusion, and coma. Cerebral edema can also lead to cerebral ischemia, herniation, and possibly death due to raised intracranial pressure. These venous collaterals dilate over several weeks and the upper body venous pressure, which is markedly elevated initially at the time of obstruction, decreases over time. During history and physical examination, emphasis should be given to the duration and severity of symptoms, history of any malignancy, previous interventions, any current or previous medications (especially antiplatelets and anticoagulants), any cardiopulmonary comorbidities, or presence of cerebral metastases. Hence lung cancer is the most common cause, accounting for almost 75% of malignant cases. Among nonsmall-cell lung cancers the most common histology found is adenocarcinoma. Of patients with small-cell lung cancer there is a higher likelihood to develop the syndrome (6% vs. This was initially treated with a 2-week course of radiotherapy but there was symptomatic recurrence. Superior vena cava successfully stented with a 16-mm diameter, 60-mm long wallstent (Boston Scientific, Inc. It is dictated by the severity of the symptoms, its likelihood of response to a particular treatment option, and the underlying malignancy treatment itself. On repeat venography, a short segment residual narrowing (arrow) is seen at the top of the stent. Radiation in particular can obscure a diagnosis in up to 42% of biopsy specimens obtained from the irradiated area after treatment. Superior venocavogram performed via an 8 French vascular sheath from a left jugular approach shows a focal area of narrowing (double headed arrow), which measured about 4 cm in length. It is usually of little benefit, however, if venous collaterals have not dilated enough. Glucocorticoid therapy (dexamethasone, 4 mg every 6 hours) is commonly prescribed, although its effects have not been formally well studied, and there are only case reports to suggest the benefit. The reported case series indicate an operative mortality rate of approximately 5% and patency rates of 80% to 90%. In patients with severe life-threatening symptoms for which urgent intervention is required, primary stenting is advocated because stenting can be done even before a tissue diagnosis is available and it provides relief rapidly and safely. But nonsmall-cell lung cancer shows poor response for chemotherapy with response seen in only 40% of cases. Secondary Stenting Secondary stenting is used for patients where primary conventional therapy failed (nonresponders) and also for those with recurrence (secondary failure) after initial response following conventional therapy or stenting itself. Brain metastasis is a relative contraindication because of the increased risk of cerebral hemorrhage when on anticoagulants after stenting. They may be classified into two categories: self-expanding stents and balloon-expanding stents. The self-expanding stents, once released, continue to push radially outward against the stenosis until they reach their designed size. The open structure of the stent does not cause obstruction to the collateral side vessels bridged by the stent like the azygos vein, but it can potentially increase the stent occlusion rate due to tumor ingrowth and restenosis. Due to its short length, large diameter, and rigidity, multiple insertions of the Z-stent for long stenoses may represent a technical challenge, especially along tortuous venous anatomy. These stents also show high levels of fatigue and stent fractures with long-term use. Unlike the Gianturco Z-stents, the nitinol self-expanding stents and wallstent stents (made of Elgiloy) are flexible, are easy to insert, and have a great efficacy in long stenoses. The main disadvantage of the wallstent is its weaker radial strength in larger diameters, its unpredictable length of foreshortening (usually 20% but can go up to 50%) when expanded, and its tendency to migrate postdeployment from the narrowest point to the normal portion of the vein if not correctly centered on the stenosis. Almost all other self-expanding stents are also flexible, like the wallstent, because they are made of nitinol (nickel titanium compound) mesh with super elasticity that allows the stent to expand to its designed size without any shortening of the length. When the delivery catheter is positioned within the stenosis, the balloon is inflated, causing the stent to expand to a desired diameter; it is therefore less likely to cause perforation. Balloon-expandable stents also allow staged dilatation of the stenosis to a larger diameter. In general, the disadvantage of the balloon-expanded stents is their relative rigidity once released and expanded, which may lead to extrinsic deformation without elastic recovery, reocclusion, or stent fracture. Overall there appears to be no significant difference in outcomes in the literature between the three general categories of stents detailed previously- the self-expanding stainless steel Gianturco Z-stent, balloon-expandable stents, and the self-expanding stents made of superelastic metals. Also, comparative studies have shown increased incidence of occlusions and complications (including mutual obstruction of stents) following bilateral stenting. The choice of using covered or bare stents is based on the type of the tumor, type of obstruction (extrinsic compression vs. Malignancies like lung cancer have a higher incidence of venous invasion with resultant tumor thrombus when compared with lymphoma or metastatic mediastinal nodal disease. In cases of significant flow through the azygos vein, however, it is beneficial to use stents like the Gianturco stents, which have an open structure to bridge the azygos vein orifice. Access from the basilic, jugular, subclavian, or femoral veins is used, depending on disease extent. However, the subclavian access route when compared with others is supposedly the more advantageous and simpler route despite a slight increased risk of pneumothorax. After crossing the obstruction, a standard hydrophilic guide wire is exchanged for a 180- or 260-cm stiff guide wire. In case of strictures, predilatation to allow passage of the stent delivery system may be necessary. However, it is vital to confirm luminal reentry prior to balloon dilatation or stenting. Balloon dilatation should always be performed with caution, however, because this may result in catastrophic venous rupture. Of the 17 deaths 7 (41%) were attributed to severe hemorrhage: 2 cerebral, 3 pulmonary, and 2 unspecified sites. Four deaths (23%) were attributed to cardiac events: two arrhythmias, one myocardial infarction, and one tamponade. One death (6%) was due to documented pulmonary embolism, and in two cases (12%) cause of death was unknown. Fatal hemorrhages were mostly attributed to thrombolytic therapy administration, including streptokinase, urokinase, and tissue plasminogen activator, which were used to dissolve intracaval clot prior to stent insertion. The predisposing factors for stent migration are poor patient selection, inaccurate vessel measurement, inadequate sizing of the stents, inaccurate positioning of the stents, and cardiac motion. These factors should be considered prior to stent deployment to minimize or prevent stent migration. This can lead to acute pulmonary edema, which can be fatal in patients with underlying coronary artery disease and heart failure. Anticoagulation is recommended following stent placement but optimal duration and methods of anticoagulation therapy remain controversial. The main drawback of this technique is that it may increase the risk of perforation of the adjacent aorta and pericardium due to embedment of sharp stent filaments into the vessel wall at the flared ends. Stenting is the treatment of choice for patients with acute life-threatening symptoms. Also outcomes and complications compare very favorably with standard therapies (such as chemotherapy and radiotherapy); therefore, it has been advocated as the first-line treatment option. Prevalence and characteristics of pleural effusions in superior vena cava syndrome. Treatment of malignant superior vena cava obstruction: metal stents or radiation therapy. Superior vena cava syndrome induced by bronchogenic carcinoma: is this an oncological emergency Clinical outcome of stenting in superior vena cava syndrome associated with malignant tumors: comparison with conventional treatment. Superior vena cava obstruction in small cell bronchogenic carcinoma: clinical parameters and survival. Emergency prebiopsy radiation for mediastinal masses: impact on subsequent pathologic diagnosis and outcome. Steroids, radiotherapy, chemotherapy and stents for superior vena caval obstruction in carcinoma of the bronchus: a systematic review. Management of superior vena cava syndrome by internal jugular to femoral vein bypass. Treatment of obstruction of the superior vena cava by combination chemotherapy with and without irradiation in small-cell carcinoma of the bronchus. Small cell lung cancer with and without superior vena cava syndrome: a multivariate analysis of prognostic factors in 408 cases. Cirse guidelines: quality assurance guidelines for superior vena cava stenting in malignant disease. Stenosis of the vena cava: preliminary assessment of treatment with expandable metal stents. Stenting as first option for endovascular treatment of malignant superior vena cava syndrome. Endovascular stenting as a first choice for the palliation of superior vena cava syndrome. Endovascular treatment of superior vena cava obstruction in patients with malignancies. Endovascular stenting in neoplastic superior vena cava syndrome prior to chemotherapy or radiotherapy. Superior vena cava syndrome: treatment with catheter directed thrombolysis and endovascular stent placement. Treatment of malignant superior vena cava syndrome by endovascular stent insertion. Safety and effectiveness of vascular endoprosthesis for malignant superior vena cava syndrome. The use of the Wallstent endovascular prosthesis in the treatment of malignant obstruction of the superior vena cava. Subacute and chronic benign superior vena cava obstructions: endovascular treatment with self expanding metallic stents. Self expanding metal stents for palliative treatment of superior vena caval syndrome. Stent therapy for malignant superior vena cava syndrome: should be first line therapy or simple adjunct to radiotherapy. Vena cava and central venous stenosis: management with Palmaz-balloon-expandable intraluminal stents. Case report: migration and shortening of a self-expanding metallic stent complicating the treatment of malignant superior vena cava stenosis. Endovascular treatment of malignant superior vena cava syndrome: is bilateral wallstent placement superior to unilateral placement Malignant superior vena cava obstruction: stent placement via the subclavian route. Cardiac tamponade: a rare complication of attempted stenting in malignant superior vena cava obstruction. Gianturco-Rosch expandable Z-stents in the treatment of superior vena cava syndrome. Long term results of endovascular stent placement in the superior caval venous system. Use of the Wallstent in the venous system including haemodialysis-related stenosis.
The investigators found a correlation between advancing obstruction pattern and the presence of symptoms keratin smoothing treatment generic 50 mg naltrexone otc. Type I is described as up to a 90% stenosis of the superior vena cava with antegrade flow through the azygos vein medicine 7767 purchase naltrexone online pills. Extrinsic compression is the result of mass effect from adjacent structures symptoms 37 weeks pregnant naltrexone 50 mg buy cheap, such as arterial aneurysm or benign masses medications hydroxyzine purchase naltrexone visa. Mediastinitis medicine bg naltrexone 50 mg purchase, either infectious or inflammatory, results in compressive forces as the surrounding fibrosis retracts and collapses the vein lumen. Intrinsic stenosis is the result of intimal trauma and pericatheter thrombus formation, which leads to smooth muscle ingrowth and the development Table 91. The process of thrombus formation further inflames and then scars the vein, resulting in worsening stenosis or occlusion. The obstruction of the superior vena cava leads to impaired venous return from the head and upper extremities. Clinical symptoms include headache, fatigue, dyspnea, cough, dysphagia, dizziness, impaired vision, nausea, syncope, and coma. In the Kishi scoring system grades were assigned to a series of clinical signs and symptoms. By summing the highest grade from each category a score was derived that was used to establish disease severity and evaluate treatment outcome. The primary treatment for malignant superior vena cava syndrome is either external radiation therapy or endovascular intervention with balloon angioplasty or stent placement. Patients with benign causes of superior vena cava syndrome tend to be comparatively younger with longer life expectancies. Initial therapy for superior vena cava obstruction of benign etiology involves anticoagulation to prevent or limit thrombus formation. Indeed some authors have concluded that endovascular intervention is now the first-line treatment for benign superior vena cava syndrome. Early use of thrombolysis for superior vena cava syndrome was focused on occlusions induced by pacemaker wires. Shortly after this initial publication, several other authors published early accounts of balloon angioplasty of the superior vena cava. By drawing on the clinical history, noninvasive imaging findings, and venographic appearance, the interventionist must determine if the vein is stenotic but patent or occluded. B vein is occluded, it is necessary to recanalize the vein because guide wire access across the lesion is fundamental to further intervention. If the vein is occluded with fresh thrombus, the guide wire typically passes without difficulty and the interventionist can proceed with thrombolysis. After excluding relevant contraindications to thrombolytic therapy (bleeding disorder, pregnancy, primary or metastatic disease of the brain or spinal cord, recent surgery, gastrointestinal bleeding, etc. First, by lysing the acute thrombus there is less potential for it to embolize during subsequent balloon angioplasty and stent placement. Second, following a course of thrombolysis, the underlying stenosis or occlusion is often shorter than had been previously appreciated and may require fewer or shorter stents. Recanalization techniques involve using various guide wires and support catheter to cross the occlusion, followed by balloon angioplasty to open the channel in preparation for balloon angioplasty and/or stent placement. In these situations, sharp recanalization can be attempted using a Colapinto needle or other such device. Postangioplasty venogram with restored patency of the previously occluded vein segment. Left arm venogram demonstrates left innominate stenosis secondary to cardiac defibrillator leads. Completion venogram following balloon angioplasty and stent placement documents restored patency of the vein segment. Heparin anticoagulation during the procedure is favored by some, although its use is not universal. By using this technique an inadvertently dislodged stent is constrained by the delivery wire and unable to travel into the heart. When faced with a stenosis or obstruction that involves the superior vena cava as well as both innominate veins, recent publications have indicated that only the superior vena cava and one innominate vein need to be revascularized to achieve clinical benefit. In comparative studies, unilateral innominate vein revascularization is favored because there is shorter procedure time, fewer complications, and less reocclusion. Removal of the pacer wire can be clinically undesirable or technically impossible. One frequently cited concern with the intervention in this setting is the possibility of a harmful interaction between the metallic stent and the pacer wire. First, pacemaker wires are well insulated by a coating of silicone rubber or polyurethane. Second, a pacemaker wire that has incited a venous stenosis is well embedded within the vessel wall and likely has no exposure to the lumen in which the stent is implanted. Whereas the reported primary patency rates of endovascular therapy are in the range of 57% to 79%, the primary assisted or secondary patency to 3 years is 96% to 100%. Major complications include stent migration, pericardial tamponade, superior vena cava rupture, pulmonary embolism, and death. Stenosis or occlusion of the superior vena cava and innominate veins can result in substantial morbidity. Whereas in the past, endovascular techniques had limited application in the treatment of superior vena cava of benign etiology, presently endovascular therapies are considered by many to be first-line treatments. Treatment option including thrombolysis, balloon angioplasty, and stent placement are often performed in conjunction with one and others. Endovascular treatment of superior vena cava syndrome results in similar outcomes as open surgical repair with relatively few complications. Open surgical and endovascular treatment of superior vena cava syndrome caused by nonmalignant disease. Impact of short-term hemodialysis catheters on the central veins: a catheter venographic study. Jugular vein catheter placement: histologic features and development of catheter-related (fibrin) sheaths in a swine model. Late symptomatic venous stenosis in three hemodialysis patients without previous central venous catheters. The role of venography and surgery in the management of patients with superior vena cava obstruction. Reconstruction of the vena cava and of its primary tributaries: a preliminary report. Transluminal angioplasty of superior vena cava obstruction in a 15-month-old child. Intravascular stents in the management of acute superior vena cava obstruction of benign etiology. Recanalization of occluded veins to provide access for central catheter placement. Subacute and chronic benign superior vena cava obstructions: endovascular treatment with self-expanding metallic stents. Treatment of superior vena cava obstruction secondary to pacemaker wires with balloon venoplasty and insertion of metallic stents. Permanent pacemakerinduced superior vena cava syndrome: successful treatment by endovascular stent. Acute occlusions have a high probability of producing bilateral lower extremity edema, abdominal and lower extremity discomfort or "heaviness," and, occasionally, ascites. With acute occlusions at or above the hepatic vein inflow, insufficient venous return to the heart can lead to fatal arrhythmias. Over time, collaterals develop gradually, reducing symptoms, frequently within 1 to 2 weeks. Note that many of these collateral channels can develop with reversed flow in patients with cirrhosis or portal hypertension, so the differential diagnosis can be quite broad based on imaging findings alone. Once the acute thrombus has been resolved, treating the underlying cause of the obstruction becomes an important consideration. For malignant direct vascular invasion or extrinsic compression from hepatic metastases, chemotherapy, radiation therapy, and surgical decompression can be considered; however, response times tend to be delayed. As one would expect, patients with short segment stenosis and acute onset of symptoms tend to respond better than the converse. Long segment occlusions with multiple collateral channels are likely to have a lower technical success rate. Contraindications to treatment would include inability to obtain stable wire access across the lesion and inability to move obstruction lesion with angioplasty or stenting devices. In short, a combination of both pharmacologic and mechanical thrombectomy agents can be used to disrupt the underlying thrombus and help establish inline venous return to the right atrium. Identifying the etiology of the underlying venous obstruction (or cause of the thrombus formation) will lead to optimum treatment options. Angioplasty, combined angioplasty and stenting, or primary stenting are endovascular treatment options for this lesion. For the most part, stenting either with or without prestent angioplasty is the most likely endovascular treatment option to have the most beneficial symptom control as well as long-term patency data. Specifically, if symptoms trend toward significant improvement with anticoagulation alone, medical management may be appropriate. Alternatively, surgical interventions, such as venous bypass or tumor debulking, may also assist in symptom control without a need for interventional techniques. Without adequate comparative studies evaluating outcomes, the choice between medical management, endovascular techniques, and surgical debulking/bypass options is usually made on a case-by-case basis (primarily based on individual risk versus potential benefit). In most cases, these symptoms are not emergent, allowing time for appropriate evaluation and planning. In lesions near the right atrium at the hepatic venous confluence, the "landing zone" of the stents may be limited. Choosing a stent with enough radial force at the diameter and lengths needed (15- to 30-mm diameter, approximately 5- to 15-cm length) can be difficult. Frequently, a diagnostic venogram prior to stent placement allows better evaluation of the length and location of the narrowing. Although no clearly defined value for a hemodynamically significant gradient in the venous system has been established, commonly stented symptomatic lesions in our practice have minimum pressure gradients in the 5- to 7-mm Hg range (unpublished data). Historically, a self-expanding stent with a strong radial force that is tolerant of considerable respiratory and cardiac pressure and diameter fluctuations over the remaining lifetime of the patient is required. On some occasions, a balloon-expandable stent may be required to reinforce the self-expandable device. Although stenting across the renal vein or hepatic vein inflow is generally avoided, there are limited data to suggest that doing so would result in a suboptimal result. Stent diameters are frequently oversized by 15% to 40% depending on the extent of the stricture and adjacent vasculature. The "oversize" component helps reduce the risk of stent migration into the right atrium, which can result in a potentially fatal arrhythmia. The length of stent chosen should be sufficient to cross the lesion in its entirety with limited extension across major inflow veins. Inferior vena cavagram demonstrates extrinsic compression from multiple hepatic metastases. Both internal jugular vein and common femoral vein access were required for evaluation of the lesion in preparation for treatment. After angioplasty and placement of a balloonexpandable stent, no flow through the collateral channels was identified. The initial inferior venacavagram demonstrates a long segment, intrahepatic stenosis with filling of right paralumbar collateral vessels. Oblique image helps define the length of compression and extent of luminal narrowing. Bilateral femoral access with measurement catheters assists in selecting the appropriate length and diameter of stent. Having a couple diameters and lengths to choose from at the time of the procedure is advisable because noninvasive imaging can occasionally misrepresent some key measurements. Planning appropriate access (femoral vein or internal jugular vein, or both) is an important component to success and is frequently operator dependent. Factors include both the diameter of the anticipated devices as well as factors involved in deployment. Large-diameter sheaths (10 to 16 French) are occasionally required to deliver the appropriate stents and any potential required angioplasty devices. A femoral approach with these devices may give the operator a slight advantage in the accurate delivery of the device at the cavoatrial junction. Pressure measurements across the lesion may help establish an appropriate endpoint after stent placement because there are limited data defining a hemodynamically significant stenosis in the venous system. With external compression lesions, a soft floppy tip wire is frequently all that is required to cross the lesion. With more chronic intravascular obstructions, a hydrophilic, steerable wire may be required. Exchange for a sturdy, "working wire" is recommended for the delivery of the large-diameter stents and angioplasty balloons. Advancing the dilator and sheath over the wire across the lesion (or "Dottering" the lesion) may help determine if prestent angioplasty may be required. If the delivery sheath cannot be safely advanced across the lesion over the wire, angioplasty with an appropriate diameter balloon (8 to 14 mm) may be of assistance. Once a delivery sheath can be positioned safely across the narrowing, a controlled, steady, and accurate deployment of the stent can be attempted. Angiography during the initial stent positioning, just prior to deployment, either via the delivery sheath or an alternative access site is frequently useful in accurate deployment of the stent.
Anticoagulation during the procedure is important to prevent immediate in-stent thrombosis medicine on time generic 50 mg naltrexone mastercard. For midthigh punctures treatment management company best naltrexone 50 mg, use of a sealing device treatment associates buy naltrexone 50 mg cheap, such as VasoSeal treatment arthritis naltrexone 50 mg buy without prescription, has been advocated treatment quad strain cheap naltrexone online amex. The patient should be administered a dose of lowmolecular-weight heparin or fondaparinux prior to departure from the recovery room to maintain therapeutic levels of anticoagulation. Whenever there is a suspicion that the catheter is not within the vessel lumen, a small injection of contrast should be performed and, if necessary, the catheter retracted until the vessel lumen is again seen. Because of the low pressure of the venous system, these false passages usually do not result in significant bleeding. If persistent bleeding is seen, placement of a bare metal stent is usually adequate to seal the venotomy. Adequate flow should be seen in both inflow and outflow vessels, although in the setting of damaged inflow vessels, the sheath itself may occlude the vessel and give the appearance of poor inflow. If an elevated venous pressure gradient was observed on initial venography, repeat pressure measurements should be obtained to assess for improvement. Raju and Neglen have published extensively from their prospective database of iliac vein stent placements dating back to 1997. In one study of 982 limbs in 870 patients stented for chronic, nonmalignant obstruction, they found primary, assisted primary, and secondary patency rates of 79%, 100%, and 100%, respectively, at 6 years for patients with nonthrombotic primary vein obstruction and 57%, 80%, and 86%, respectively, for patients with post-thrombotic obstruction. Factors associated with early or late stent occlusion included thrombotic etiology of lesions, long lesions requiring multiple stents, extension into the common femoral vein, and high degree of stenosis or total vein occlusion prior to stenting. The percentage of limbs free of pain improved from 24% before treatment to 62% 5 years after treatment; the percentage of limbs free from swelling improved from 18% before treatment to 32% 5 years after treatment. Improvement in pain was seen in 78% and complete pain relief was seen in 71% of patients at 5 years; for swelling, 55% of patients saw improvement and 36% had complete relief in 5 years. They experienced five complications: two stent migrations, one superficial femoral artery tear treated percutaneously, and two cases of contrast extravasation during recanalization. In the subset of patients followed greater than 4 years, the primary patency rate at 4 years was 78%, the assisted-primary patency rate was 88%, and the secondary patency rate was 90%. They attributed restenoses to underestimation of the length of vein to be stented or, in one case, inadequate overlap between stents. Objective measurements, such as calf and thigh circumference at reproducible locations, should be documented both before and after therapy. Response to therapy can be quantified using severity scores based on physician assessment and patient quality-of-life surveys. Aggressive attempts to recanalize may result in the wire creating false passages outside the vessel, making it even more difficult to recanalize the true vessel lumen. In one series of 167 limbs in 159 patients, the authors had a technical success rate of 83%. Occasionally, on completion venography after stent deployment, there is poor flow within the stents. This can be caused by inadequate coverage of diseased vein by the stents; further extension of the stents may be necessary. This may be caused by inadequate anticoagulation of the patient during the procedure. The thrombus can be treated with balloon angioplasty and/or rheolytic thrombectomy. Potential causes include inadequate anticoagulation in the immediate postprocedural period, failure to stent the full extent of diseased vein, or mechanical stresses on the stent (such as when it is placed across the inguinal ligament). Extending the stent or placing an overlapping stent to increase radial force may be necessary. In cases refractory to endovascular management, consideration can be given to open surgical venovenous bypass grafting. Venography can be performed in cases in which noninvasive imaging is inconclusive or when further intervention is warranted. Patients should be aggressively anticoagulated and maintained at therapeutic levels from the immediate perioperative period through the first 3 to 6 months after stent placement. The techniques and materials used to perform these procedures are commonly found in the interventional radiology armamentarium, making this a procedure that can easily be widely adopted. Additionally, some patients with long-segment chronic occlusion cannot be successfully recanalized. Stent surface treatments to prevent in-stent thrombosis could assist in maintaining primary patency. No randomized controlled trials have been performed to compare outcomes after venography with and without intervention. Comparison with surgical management is also difficult because the advent of endovascular therapy has diminished the role of surgical venous bypasses, which are technically challenging and not universally available. Bypass surgeries now tend to be reserved for patients who have failed endovascular management or who are undergoing concurrent surgery for resection of malignancy. The contralateral saphenous vein serves as the bypass conduit, connecting the common femoral veins through a suprapubic subcutaneous tunnel. If saphenous vein is not available, expanded polytetrafluoroethylene graft can be used, although the patency rates are much lower. A temporary arteriovenous fistula is occasionally placed to maintain graft patency. An analysis of 412 operations in nine series demonstrated a crude patency rate of 70% to 85%, although duration of follow-up and use of objective measures of graft patency varied between the studies. One group found the 2-year primary and secondary patency rates of 13 bypass grafts (5 iliocaval and 8 femorocaval) to be 38% and 54%, respectively. Importance, etiology, and diagnosis of chronic proximal venous outflow obstruction. Endovascular management of chronic disabling ilio-caval obstructive lesions: long-term results. Venous function assessed during a 5 year period after acute ilio-femoral venous thrombosis treated with anticoagulation. Relationship between changes in the deep venous system and the development of the postthrombotic syndrome after an acute episode of lower limb deep vein thrombosis: a one- to six-year follow-up. High prevalence of nonthrombotic iliac vein lesions in chronic venous disease: a permissive role in pathogenicity. Use of compression stockings in chronic venous disease: patient compliance and efficacy. Venous leg ulcer: a meta-analysis of adjunctive therapy with micronized purified flavonoid fraction. Venous recanalization by metallic stents after failure of balloon angioplasty or surgery: four-year experience. Revision of the venous clinical severity score: venous outcomes consensus statement: special communication of the American Venous Forum Ad Hoc Outcomes Working Group. Whereas endovascular management of iliocaval occlusion is effective in many of those patients, the outcomes are less favorable than those for patients with primary vein obstruction. Relationship between clinical classification of chronic venous disease and patient-reported quality of life: results from an international cohort study. Stenting of the venous outflow in chronic venous disease: long-term stent-related outcome, clinical, and hemodynamic result. Surgical reconstruction of iliofemoral veins and the inferior vena cava for nonmalignant occlusive disease. Initially described as a cause of intestinal infarction by Elliot in 1895,1 mesenteric venous thrombosis is responsible for 5% to 15% of all mesenteric ischemic events. Nonspecific symptomatology delays diagnosis and contributes to the poor outcomes associated with the condition, although improved imaging techniques have enabled more rapid recognition and detection. Additionally, improved understanding of underlying disease mechanisms has improved both treatment and outcomes. Despite a vague clinical presentation, the prompt recognition and aggressive treatment of this condition is necessitated by high morbidity and mortality rates related to intestinal infarction, bowel perforation, and high recurrence rates. Oral contraceptive use is responsible for 4% to 5% of total mesenteric venous thrombotic events, a higher percentage of cases occurring in young women. Direct Injury Direct injury is recognized as a predisposing condition for mesenteric venous thrombosis. These injuries include pancreatitis, inflammatory bowel disease, abdominal trauma, and postsurgical trauma. Primary mesenteric venous thrombosis is defined as spontaneous, idiopathic thrombosis of the mesenteric veins and accounts for approximately 25% of cases. Local Venous Stasis Conditions causing venous stasis in the mesenteric system include cirrhosis, portal hypertension, severe congestive heart failure, and morbid obesity. These patients must be managed with lifelong anticoagulation to prevent recurrence. Additional hematologic hypercoagulable states associated with mesenteric venous thrombosis include polycythemia vera, thrombocytosis, hyperfibrinogenemia, paroxysmal nocturnal hemoglobinuria, and myeloproliferative states. The location of acute thrombus formation can be an important predictor of the underlying cause of mesenteric venous thrombosis. Intra-abdominal causes, such as splenectomy and pancreatitis, initiate thrombus formation in the large veins and spread peripherally. Hypercoagulable states often lead to thrombus formation in the vasa recta, arcuate veins, and venous arcades and progress to involve the larger veins. Even after the venous obstruction has been relieved, arterial vasoconstriction can persist, adequate to cause intestinal infarction and arterial thrombosis. Mesenteric ischemia usually presents with generalized midabdominal pain not explained by physical findings and is often accompanied by vomiting, abdominal distention, and constipation. In these cases, onset of symptoms is sudden with colicky and severe pain, out of proportion to abdominal findings on physical exam. More than half of patients report other nonspecific symptoms, such as nausea/vomiting, anorexia, and constipation with or without bloody diarrhea. In those patients with subacute mesenteric venous thrombosis, abdominal pain is the major finding without bowel infarction or variceal hemorrhage. Patients with chronic mesenteric venous thrombosis have extensive venous collateral circulation and therefore less abdominal pain. The diagnosis is usually one of exclusion, after more common causes of abdominal pain, such as cholecystitis, pancreatitis, gastroesophageal reflux, and gastric disorders, have been explored. These cases may demonstrate weight loss, food avoidance, and vague postprandial abdominal pain. The differential diagnosis for acute mesenteric venous thrombosis is lengthy and includes both intravascular and extravascular causes. Arterial causes include embolic disease, occlusive/nononcclusive atherosclerotic disease, dissecting aortic aneurysm, hypoperfusion, and disseminated intravascular coagulation. Extravasular causes include incarcerated hernias, small bowel obstruction, volvulus, intussusception, and adhesive bands/scars. Patients with subacute or chronic mesenteric venous occlusion frequently have thrombosis of the portal and/or splenic veins, sparing the vasa recta and venous arcades. These patients present less acutely because venous collaterals provide sufficient venous drainage. Whatever the mechanism, occlusion of the mesenteric veins causes inadequate venous drainage of the bowel. Ischemic bowel becomes edematous and thickened with intramural hemorrhage, leading to rapid loss of fluid into the bowel lumen, mesentery, and peritoneal cavity. The increased venous pressure results in hemorrhage, perforation, and peritonitis. Metabolic acidosis or elevated lactate levels are late findings associated with bowel infarction. Findings on abdominal X-rays include dilated loops of small bowel with air-fluid levels indicative of small bowel obstruction or ileus, thickened loops of bowel or "thumbprinting" indicative of bowel wall edema, and pneumatosis intestinalis. B conditions including portal hypertension and gallbladder disease, portomesenteric thrombosis may be an incidental finding. This modality has several limitations: its operator-dependent nature, limited visualization due to overlying bowel gas or large patient body habitus, and sensitivity to detect slow flow. The mesenteric veins may have thick, enhancing walls and multivein occlusion is common. Bowel wall dilatation is a common manifestation reflecting absence of peristalsis secondary to both arterial and venous perfusion abnormalities. Associated findings include stranding in the mesentery, collateral circulation, and ascites. Pneumatosis intestinalis, portomesenteric gas, and free intraperitoneal air are late findings of transmural infarction and perforation. In patients with chronic mesenteric venous occlusion, cavernous transformation of the portal vein, abundant venous collaterals, gastroesophageal varices, and ascites can be identified in the absence of intestinal ischemia. Additionally, "ectopic" varices in the small intestine, colon, and rectum can be diagnosed. Because of the advances in noninvasive imaging, conventional angiography is not routinely used for the diagnosis of mesenteric venous thrombosis. Instead, it is used for equivocal cases and in conjunction with transcatheter therapies. The primary aim of treatment is to avoid bowel infarction, peritonitis, and ischemia. In the absence of ongoing ischemia, oral anticoagulation with warfarin should be started and continued for 3 to 6 months or lifetime, depending on the underlying cause. The use of anticoagulants in patients with chronic mesenteric venous ischemia is controversial; although beneficial in preventing new thromboses, it carries the risk of exacerbating variceal hemorrhage.
50 mg naltrexone buy with mastercard. FDA 'Z pack' antibiotics could cause deadly heart problems.
References
- Marinho S, Simpson A, So?derstro?m L, et al. Quantification of atopy and the probability of rhinitis in preschool children: a population-based birth cohort study. Allergy 2007; 62: 1379-1386.
- Nataatmadja M, West M, West J, et al: Abnormal extracellular matrix protein transport associated with increased apoptosis of vascular smooth muscle cells in Marfan syndrome and bicuspid aortic valve thoracic aortic aneurysm, Circulation 108(Suppl 1): II329, 2003.
- Rodgers A, Allie-Hamdulay S, Jackson G: Therapeutic action of citrate in urolithiasis explained by chemical speciation: increase in pH is the determinant factor, Nephrol Dial Transplant 21:361n369, 2006.
- Wiener-Kronish JP, Albertine KH, Matthay MA. Differential responses of the endothelial and epithelial barriers of the lung in sheep to Escherichia coli endotoxin. J Clin Invest 1991;88:864-75.
