
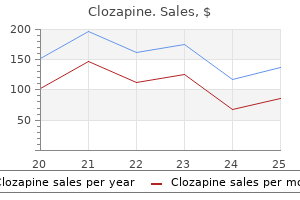
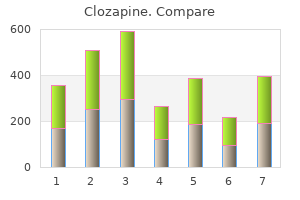
Clozapine
| Contato
Página Inicial
Michele P Mohajer BM BS FRCOG MD
- Consultant in Maternal and Fetal Medicine, Royal Shrewsbury
- Hospital, Shrewsbury
However depression not eating clozapine 25 mg buy cheap, the shortand long-term consequences associated with azathioprine are less well known and worth listing depression test quotev buy generic clozapine 100 mg on-line. Bone marrow suppression anxiety hypnosis buy clozapine 50 mg without prescription, with anemia anxiety 911 purchase clozapine 25 mg amex, leucopenia bipolar depression xanax discount 25 mg clozapine otc, and thrombocytopenia, is the most common laboratory abnormality. Symptomatically, nausea, vomiting, and abdominal pain may lead to cessation of therapy due to intolerance. Acute pancreatitis is rare, as is hepatoxicity, usually marked by sinusoidal obstructive syndrome or cholestasis, both of which usually respond to drug cessation. Long term, malignancy can occur, usually a lymphoma or leukemia (Kandiel et al, 2005). However, one series of patients with autoimmune hepatitis followed during 20 years showed no incidence of malignancy (Johnson et al, 1995). Allopurinol inhibits xanthine oxidase, an enzyme necessary for the breakdown of azathioprine. If this enzyme is inhibited, the half-life of azathioprine is prolonged and can be toxic, with profound bone marrow suppression. Other agents have been used for the treatment of autoimmune hepatitis, although the experience is predominantly anecdotal. Mycophenolate mofetil, another lymphocyte inhibitor used in solid-organ transplant recipients, can be substituted for azathioprine in cases of intolerable side effects, with comparable efficacy, based on a relatively limited experience. Surgery in the Patient With Autoimmune Hepatitis No special precautions need be taken in patients with autoimmune hepatitis requiring surgery. Patients with acute autoimmune hepatitis with signs of acute liver failure or cirrhosis clearly are at risk; however, indications for surgery in this setting would likely be only of an emergent nature (see Chapter 77). In the patient with well-controlled autoimmune hepatitis on maintenance immunosuppression, the risk of liver decompensation and/or a disease flare is small, as long as patients are maintained on their medications. Stress doses of corticosteroids should be considered if a patient is on corticosteroids or has recently been tapered off. If a patient is on azathioprine monotherapy preoperatively, the dose should be restarted as soon as possible postoperatively. A prolonged absence of gut motility should lead to discussion with a hepatologist regarding the need for intravenous steroids to keep the disease in remission. Routinely, a few days off immunosuppressive therapy will not lead to a disease flare; thus the potential risks and benefits associated with corticosteroids need to be carefully considered. Patients with advanced cirrhosis routinely are not surgical candidates, except in cases of emergent and/or life-threatening events requiring surgical intervention. Liver disease, especially significant fibrosis or cirrhosis, represents a significant risk factor for perioperative morbidity and mortality. Any clinically evident signs of liver dysfunction should raise concern, particularly for intraabdominal operations (see Chapter 77). Although there is no absolute consensus on risk quantification, it is clear the perioperative risk in patients with liver disease increases with the severity of liver dysfunction (The et al, 2007). These factors should clearly influence the informed consent for the patient and family. When possible, patients should be referred to a specialty liver care center where preoperative optimization should be carried out via a multidisciplinary team approach. Hepatitis Chapter 70 Chronic hepatitis: epidemiology, clinical features, and management1064. Benova L, et al: Vertical transmission of hepatitis C virus: systematic review and meta-analysis, Clin Infect Dis 59:765773, 2014. Fassio E, et al: Natural history of non-alcoholic steatohepatitis: a longitudinal study of repeat liver biopsies, Hepatology 40:820826, 2004. Report of the National Heart, Lung, and Blood Institute/American Heart Association Conference on Scientific Issues Related to Definition, Circulation 109:433438, 2004. Kandiel A, et al: Increased risk of lymphoma among inflammatory bowel disease patients treated with azathioprine and 6-mercaptopurine, Gut 54:11211125, 2005. Kumar M, et al: A randomized controlled trial of lamivudine to treat acute hepatitis B, Hepatology 45:97101, 2007. Lawitz E, et al: Sofosbuvir for previously untreated chronic hepatitis C infection, N Engl J Med 368:18781887, 2013. Morisaki A, et al: Risk factor analysis in patients with Liver cirrhosis undergoing cardiovascular operations, Ann Thorac Surg 89:811817, 2010. Piton A, et al: Factors associated with serum alanine transaminase activity in healthy subjects: consequences for the definition of normal values, for selection of blood donors and for patients with chronic hepatitis C. Saadeh S, et al: the utility of radiologic imaging in nonalcoholic fatty liver disease, Gastroenterology 123:745750, 2002. Vernon G, et al: Systematic review: the epidemiology and natural history of non-alcoholic fatty liver and non-alcoholic steatohepatitis in adults, Aliment Pharmacol Ther 34:274285, 2011. Understanding the effects of back ground liver injury on postoperative outcomes is crucial to efforts aimed at improving the safety of liver resection. Hepatic steatosis is defined as abnormal macrovesicular or microvesicular triglyceride accumulation in at least 5% of hepa tocytes diagnosed histologically or by radiologic imaging (Aly & Kleiner, 2011; Chalasani et al, 2012; Sanyal et al, 2011). In adults, the initial injury usually occurs in the perivenular (zone 3) portion of the hepatic acinus, which is the least oxygenated and most prone to freeradicalmediated injury. This is in con trast to other chronic liver diseases, in which marked portal inflammation is observed early in disease course (see Chapter 76). Bor derline steatohepatitis includes some, but not all, of these fea tures (Kleiner & Brunt, 2012). In more advanced stages, periportal fibrosis may occur with possible progression to bridging fibrosis (Kleiner & Brunt, 2012). A, Steatohepatitis with moderate steatosis, lobular inflammation (arrow), and hepatocyte ballooning degeneration (arrowheads) observed. Potential risk factors include alcohol use, elements of the metabolic syndrome, hereditary disorders, and druginduced injury, particularly from chemotherapeutics. This congestion blocks venous outflow, resulting in sinusoidal dilatation and deposition of extracellular collagen, leading to perisinusoidal fibrosis. Nodular regenerative hyperplasia is characterized by the diffuse transformation of normal hepatic parenchyma into small, regenerative nodules that compress surrounding parenchyma and can cause portal hypertension (RubbiaBrandt et al, 2010). Obesity, 36%, (Geiss et al, 2014; May et al, 2013; Ogden et al, 2014); diabetes mellitus, 8% to 10%, (Geiss et al, 2014; Selvin et al, 2014); and the metabolic syndrome, 23%, (BeltranSanchez et al, 2013) are prevalent in the United States. Relative enhancement in these conditions is less compared with simple steatosis (Bastati et al, 2014). Steatohepatitic features did not affect overall or diseasefree survival after liver resection. Recurrence-free survivaldifference statisticallysignificant Resectionand transplant Mediannot reachedvs. Other studies have noted only a relationship with irinotecan and steatohepatitis, but not steatosis (Robinson et al, 2012; Vauthey et al, 2006). A twophase mechanism by which irino tecan may cause steatohepatitis has been proposed in which components of the metabolic syndrome (especially insulin resistance) leads to excess fattyacid deposition in hepatocytes, resulting in increased production of reactive oxidation species. Studies showing that both metabolic factors and irinotecan therapy contribute to steatohepatitis, or which suggest a synergistic relationship between obesity and irinotecan in promoting steatohepatitis, support this hypothesis (Vauthey et al, 2006; Wolf et al, 2013). Finally, some reports note no association between any chemo therapeutic agent and steatohepatitis, citing only a relationship with obesity (Brouquet et al, 2009; Makowiec et al, 2011; Ryan et al, 2010). Sinusoidal obstruction, centrilobular perisinusoidal and venular fibrosis, and nodular regenerative hyperplasia have all been reported with oxaliplatin therapy (Brouquet et al, 2009; Robin son et al, 2012; RubbiaBrandt et al, 2010; Ryan et al, 2010). The pres ence of the metabolic syndrome and individual elements are associated within superficial surgicalsite infections, sepsis, acute renal failure, and cardiopulmonary morbidity and mortal ity after liver resection (Bhayani et al, 2012; Le Bian et al, 2012; A. Prospective randomized controlled trials have shown an increase in postoperative morbidity-particularly bile leak-after liver resection with preoperative chemotherapy. Several series have analyzed the impact of underlying liver injury on postoperative outcomes after partial hepatectomy (Table 71. Other studies have noted that elements associated with liver injury, such as chemotherapeutic treatment (Schwarz et al, 2013) or components of the metabolic syndrome, are associ ated with poor outcomes after liver resection. To address this issue, we performed a cohortmatched comparison of noncirrhotic patients with hepatic steatosis greater than 33% or steatohepatitis with respective matched controls with no underlying liver disease (Reddy et al, 2012). In con trast, there was no difference in any postoperative outcome between patients with hepatic steatosis and matched controls. Impor tantly, the etiology of background liver injury was not associated with hepaticrelated postoperative outcomes, indicating that hepaticrelated morbidity in patients with steatohepatitis is the result of liver injury and not from other side effects derived from factors that predispose to this injury. Our results also stress the importance of discerning between steatosis and steatohepatitis and may explain the inconsistency regarding the impact of severity of steatosis on postoperative outcomes observed in other studies (Abdalla & Vauthey, 2007; Cauchy et al, 2014). Knowledge of background hepatic injury will alter preopera tive management aimed at improving the safety of liver resec tion. A key factor in determining postopera tive outcome after resection is the volume of liver remnant as this is a surrogate for liver function. Recognition of these liver injuries may alter treatment strategies aimed at increasing anticipated liver remnant volume before liver resection with preoperative portal vein embolization or use of combination resection plus ablation as a parenchymalsparing strategy (Evrard et al, 2014; Faitot et al, 2014, Zorzi et al, 2007) (see Chapter 108). Alexander J, et al: Nonalcoholic fatty liver disease contributes to hepa tocarcinogenesis in noncirrhotic liver: a clinical and pathological study, J Gastroenterol Hepatol 28:848854, 2013. Aloia T, et al: Liver histology and surgical outcomes after preoperative chemotherapy with fluorouracil plus oxaliplatin in colorectal cancer liver metastases, J Clin Oncol 24:49834990, 2006. Angulo P, et al: Simple noninvasive systems predict longterm out comes of patients with nonalcoholic fatty liver disease, Gastroenterology 145:782789, 2013. Possible application in a population study on the metabolic syndrome, Inter Emerg Med 7(Suppl 3):S283S290, 2012. Assy N, et al: Fatty infiltration of liver in hyperlipidemic patients, Dig Dis Sci 45:19291934, 2000. Bedossa P, et al: Evidence for a role of nonalcoholic hepatitis in hepa titis C: a prospective study, Hepatology 46:380387, 2007. BeltranSanchez H, et al: Prevalence and trends of metabolic syndrome in the adult U. Berman K, et al: Hepatic and extrahepatic cancer in cirrhosis, Am J Gastroenterol 106:899906, 2011. Beymer C, et al: Prevalence and predictors of asymptomatic liver disease in patients undergoing gastric bypass surgery, Arch Surg 138: 12401244, 2003. Brouquet A, et al: Risk factors for chemotherapyassociated liver inju ries: a multivariate analysis of a group of 146 patients with colorectal metastases, Surgery 145:362371, 2009. Bugianesi E, et al: Expanding the natural history of nonalcoholic ste atohepatitis: from cryptogenic cirrhosis to hepatocellular carcinoma, Gastroenterology 123:134140, 2002. Cauchy F, et al: Surgical treatment of hepatocellular carcinoma associ ated with the metabolic syndrome, Br J Surg 100:113121, 2013. Cauchy F, et al: Metabolic syndrome and nonalcoholic fatty liver disease in liver surgery: the new scourges Chalasani N, et al: the diagnosis and management of nonalcoholic fatty liver disease: practice guideline by the American Gastroentero logical Association, American Association for the Study of Liver Diseases, and the American College of Gastroenterology, Gastroenterology 142:15921609, 2012. Doycheva I, et al: Prognostic implication of liver histology in patients with nonalcoholic fatty liver disease in diabetes, J Diabetes Complications 27:293300, 2013. Faitot F, et al: Twostage hepatectomy versus 1stage resection com bined with radiofrequency ablation for bilobar colorectal liver metas tases: a casematched analysis of surgical and oncological outcomes, Ann Surg 260:822827, 2014. Gambino R, et al: Redox balance in the pathogenesis of nonalcoholic fatty liver disease: mechanisms and therapeutic opportunities, Antioxid Redox Signal 15:13251365, 2011. Gaudio E, et al: Nonalcoholic fatty liver disease and atherosclerosis, Intern Emerg Med 7:S297S305, 2012. Gomez D, et al: Steatosis predicts postoperative morbidity following hepatic resection for colorectal metastasis, Br J Surg 94:13951402, 2007. Goossens N, Negro F: the impact of obesity and metabolic syndrome on chronic hepatitis C, Clin Liver Dis 18:147156, 2014. Hashimoto E, et al: Hepatocellular carcinoma in patients with nonal coholic steatohepatitis, J Gastroenterol 44(Suppl 19):8995, 2009. HernandezAlejandro R, et al: A comparison of survival and pathologic features of nonalcoholic steatohepatitis and hepatitis C virus patients with hepatocellular carcinoma, World J Gastroenterol 18:41454149, 2012. Jain D, et al: Steatohepatitic hepatocellular carcinoma, a morphologic indicator of associated metabolic risk factors, Arch Pathol Lab Med 137:961966, 2013. Jinjuvadia R, et al: the association between metabolic syndrome and hepatocellular carcinoma: systemic review and metaanalysis, J Clin Gastroenterol 48:172177, 2014. Kawada N, et al: Hepatocellular carcinoma arising from noncirrhotic nonalcoholic steatohepatitis, J Gastroenterol 44:11901194, 2009. Kim D, et al: Association between noninvasive fibrosis markers and mortality among adults with nonalcoholic fatty liver disease in the United States, Hepatology 57:13571365, 2013. Kishi Y, et al: Extended preoperative chemotherapy does not improved pathologic response and increases postoperative liver insufficiency after hepatic resection for colorectal liver metastases, Ann Surg Oncol 17:28702876, 2010. Lazo M, et al: Prevalence of nonalcoholic fatty liver disease in the United States: the Third National Health and Nutrition Examina tion Survey, 19881994, Am J Epidemiol 178:3845, 2013. Loomba R, et al: Association between diabetes, family history of dia betes, and risk of nonalcoholic steatohepatitis and fibrosis, Hepatology 56:943951, 2012. Makowiec F, et al: Chemotherapy, liver injury, and postoperative com plications in colorectal liver metastases, J Gastrointest Surg 15:153 164, 2011. McCormack L, et al: Hepatic steatosis is a risk factor for postoperative complications after major hepatectomy: a matched casecontrol study, Ann Surg 245:923930, 2007. McWhirter D, et al: Chemotherapy induced hepatotoxicity in meta static colorectal cancer: a review of mechanisms and outcomes, Crit Rev Oncol Hematol 88:404415, 2013. Nakano H, et al: Sinusoidal injury increases morbidity after major hepatectomy in patients with colorectal liver metastases receiving preoperative chemotherapy, Ann Surg 247:118124, 2008. Nalbantoglu I, et al: Histological features and severity of oxaliplatin induced liver injury and clinical associations, J Dig Dis 15:553560, 2014.
An important aspect of the Hassab procedure is the absence of esophageal transection and pyloroplasty mood disorder nos 2969 buy clozapine from india. Hassab suggested that portoazygous disconnection was sufficient and esophageal transection was unnecessary definition depression topographic map clozapine 100 mg order visa. The left gastric (coronary) vein is divided between ligatures depression rehab buy genuine clozapine line, that is depression gerd symptoms cheap clozapine 100 mg without prescription, the main trunk of the left gastric vein is not preserved in the Hassab procedure Hassab (1998) suggested that combined sclerotherapy or endoscopic ligation could reduce rebleeding if there were missed obliterated esophageal perforators or if devascularization at operation was incomplete mood disorder 7 year old clozapine 25 mg purchase. Several authors have practiced the Hassab procedure or modified Hassab procedure with good results. It included 174 patients operated during or after a bleed, with 39 patients operated under emergency conditions at the time of bleeding, and in 151 patients, devascularization was performed prophylactically. Hassab reported excellent results, with an in-hospital mortality of 9% for elective cases, whereas in patients operated during bleeding in an emergency setting, the mortality was 38. Although early fatal rebleeding occurred in 8 patients, there was only one late rebleeding event during follow-up. The authors reported that varices disappeared completely or improved in 91% of patients. The procedure consists of a transthoracic and an abdominal procedure performed through two separate incisions. The thoracic procedure involves extensive paraesophageal devascularization up to the inferior pulmonary vein and esophageal transection. The abdominal procedure includes splenectomy, devascularization of the abdominal esophagus and cardia, and selective vagotomy and pyloroplasty. In 29 poor-risk patients, the two parts of the operation, that is, thoracic and abdominal, were done in two stages 4 to 6 weeks apart, transthoracic being the first stage in 21 patients and transabdominal in 8 patients. The hiatus now is devascularized, and the lower esophagus is transected at the level of the diaphragm. The posterior muscular layer was left intact in the original description, and approximately 70 to 90 interrupted sutures are applied to occlude the divided varices. Because of the high risk of an esophageal anastomotic leak, esophageal transection is not done in the patients who very recently have had extensive endoscopic therapy in the form of injection sclerotherapy or variceal band ligation. Sugiura-Futagawa Operation: Abdominal Procedure Laparotomy is performed through an upper midline incision with left lateral extension. The first step is to perform a splenectomy, followed by devascularization of the abdominal part of the esophagus and cardia, followed by the greater curvature. Lesser curvature devascularization follows, and the cardioesophageal branches of left gastric vessels (but not the main left gastric) are ligated and divided. The length of devascularization is approximately 7 cm of the lesser curvature (two-thirds of lesser curvature); the lower extent of the devascularization reaches approximately the midpoint of the lesser curvature, preserving only two descending branches of the left gastric artery. The esophagus and cardia are completely mobilized and freed from the adjacent structures. Devascularization is facilitated by division of anterior vagus nerve, and for this reason, a pyloroplasty is performed. Thus, conceptually, the Sugiura procedure combines splenectomy with esophagogastric devascularization, which destroys (ligates and divides) the intraesophageal portocaval shunt but preserves periesophageal portacaval shunt, as only the perforating veins draining the esophagogastric varices are divided, and the periesophageal plexus of veins that connect the coronary venous system are preserved. The splenectomy does decrease the total portal blood flow and thereby pressure, but not to the extent of a total shunt procedure and therefore lessens the risk of hepatic decompensation. Sugiura and Futagawa (1973) reported the disappearance of varices in 71 of 73 survivors (97%). In the larger experience reported by Sugiura and Futagawa in 1984, of the 671 patients who underwent the procedure, 203 (30%) had prophylactic, 363 (54%) had elective, and 105 (16%) had an emergency procedure. In patients with cirrhosis, ChildPugh statusbased mortality was 0% for 244 Child-Pugh class A patients, 2% for 251 class B patients, and 16% for 176 class C patients. The overall survival rate was 46% for group C compared with 86% and 81% for group A and B, respectively. Late deaths were due to hepatic failure and hepatocellular carcinoma and not due to variceal bleeding. The 10 year actuarial survival rate in patients with cirrhosis was 55% in emergency cases, 72% in prophylactic cases, and 72% in Sugiura-Futagawa Operation: Thoracic Procedure In the thoracic component of the operation, a left lateral thoracotomy is performed in the sixth intercostal space, and the mediastinal pleura is incised. Care is taken to preserve the dilated collateral veins running parallel to the esophagus, and only the shunts (vascular channels) from these to the esophagus are completely ligated and divided. These perforating veins must be completely and systematically ligated and divided to devascularize the esophagus. Hepatic Cirrhosis, Portal Hypertension, and Hepatic Failure Chapter 84 Portal hypertensive bleeding: operative devascularization 1211 elective cases. The corresponding survival rates in patients without cirrhosis were 90% in emergency cases, 96% in prophylactic cases, and 95% in elective cases. The Sugiura procedure was simplified by several surgeons using an exclusive abdominal approach and stapled transection of the esophagus, achieving similar results to the combined thoracic and abdominal approach (Ginsberg et al, 1982; Inokuchi, 1985; Peracchia et al, 1980). The standard modifications include completing the entire procedure by the abdominal approach, esophagogastric devascularization, preserving the coronary vein and the longitudinal periesophageal collateral veins, and esophageal transection using staplers. In 1974, Van Kemmel was the first to use a circular stapling device for performing esophageal transection. Peracchia and colleagues, in 1980, modified the technique to include splenectomy, devascularization of the gastric corpus, fundus, and distal esophagus; resection-anastomosis of the lower esophagus using a circular stapling device; selective vagotomy with pyloromyotomy; and antireflux cardioplasty done through a laparotomy. All the modifications suggested and practiced have at the core the above-mentioned principles, with variations being inclusion or exclusion of esophageal transection, splenectomy, vagal preservation, and antireflux surgery (Dong et al, 2004; Ginsberg et al, 1982; Hidalgo Huerta et al, 1983; Johnson et al, 2006; Mariette et al, 1994; Mercado, 1993; Orozco et al, 1994; Shah et al, 1999; Umeyama et al, 1983; Yamamoto et al, 1976). Johnson and colleagues (2006) performed a retrospective analysis of devascularization with or without esophageal transection combined with salvage endotherapy and pharmacotherapy for control of a variceal bleeding and concluded that devascularization without esophageal stapler transection is a safe and effective procedure for adequate control of variceal bleeding, with similar results and less morbidity when compared with devascularization with esophageal transection in both patients with and without cirrhosis. The main vagal trunk is preserved; highly selective vagotomy is performed, and therefore no drainage procedure is necessary. Alternatively, stapling of the anterior and posterior walls can be achieved by using thoracoabdominal stapler (without a cutter) introduced via enterotomy on either side. Previous sclerotherapy induces periesophageal fibrosis, which poses a threat of injuring the esophageal wall during devascularization. When the esophagus is inflamed because of multiple sessions of sclerotherapy, especially in the acute setting, the stapling is done just below the gastroesophageal junction (Chaudhary & Aranya, 1991). Occasionally, fundic resection is needed for bleeding from large fundic varices (Han et al, 2004; Lee et al, 2009). Sometimes, early gastrotomy to oversew gastric varices is needed in acutely bleeding gastric varices. When the Sengstaken-Blakemore tube is used for temporary control of bleeding, we first perform the esophagogastric devascularization and splenic artery ligation without deflating the tube and later proceed to splenectomy. Placement of a feeding jejunostomy in patients with esophageal transection or stapling permits early institution of enteral nutrition postoperatively. A gastrograffin swallow is done around the seventh postoperative day, after which oral alimentation is resumed. Kitano and colleagues (1994) first described laparoscopic assisted devascularization in seven patients with cirrhosis with bleeding esophageal varices. Manzano-Trovamala and colleagues in 1996 performed the totally laparoscopic devascularization. Several authors have subsequently performed a devascularization procedure by a minimal invasive approach with several modifications, with very good results (Akahoshi et al, 2014; Cheng et al, 2014; Danis et al, 2004; Helmy et al, 2003; Wang et al, 2008, 2015; Yamamoto et al, 2006; Zhao et al, 2013). Devascularization through a single port has also been described (Jing et al, 2013; Wu et al, 2013; Xu et al, 2014). Jiang and colleagues (2009) compared open and laparoscopic splenectomy with azygoportal devascularization and concluded that the laparoscopic procedure was a safe and effective method in carefully selected patients. Laparoscopic Devascularization: Procedure the patient lies supine strapped to the table to allow for changes in position. The patient is placed in the reverse Trendelenberg position, and the splenic hilum is exposed by dividing the splenogastric, gastrocolic, and splenocolic ligaments. The remaining attachments (splenophrenic and splenorenal) are divided so that the spleen now has only the hilar attachments left. The posterior gastric vein, the main branch of gastric coronary vein, and the left gastric artery are identified and divided. The esophagus is pulled inferiorly, and the lower 6 to 10 cm of the esophagus is devascularized. The spleen is placed in a retrieval bag, morcellated, and extracted from the 12 mm trocar. The head of the circular stapler has been closed, and the esophageal anastomosishasbeencompleted. In a meta-analysis of these data, the hemodynamic parameters showed a significant decrease of portal vein pressure, portal vein diameter, and free portal pressure in the combined group compared with the devascularization group. The authors claim that combined procedures integrate the advantages of shunt surgery with those of devascularization, including maintaining the normal anatomic structure of the portal vein (Yin et al, 2013). However, we feel that it defeats the very reason of performing a devascularization procedure instead of shunting, because devascularization is often performed in patients in whom a shunt is either not feasible or considered to be unsafe. At best, it could imply that patients chosen for a shunt procedure may benefit by addition of limited devascularization. A more radical approach, in form of partial or total esophagogastrectomy with jejunal or colonic conduit for bypass, has been suggested by some authors (Habif, 1950; Lynn, 1971; Orloff et al, 1994; Schafer et al, 1950). However, such extensive complex surgical procedures require multiple anastomosis, and therefore, are not popular in these often unstable patients. These are useful only as the last resort in patients with extrahepatic portal obstruction with unshuntable veins and a failed devascularization procedure. The overall operative mortality rate of the Hassab procedure in Egypt (Hassab, 1967) was reported as 12. When the devascularization operation is used as an emergency procedure, the operative mortality increases significantly to 38. In patients with cirrhosis, the Child-Pugh statusbased mortality was 0 % for 244 Child-Pugh class A patients, 2% for 251 class B patients, and 16% for 176 class C patients. In the Western series, the operative mortality of the operation performed as an emergency procedure for variceal bleeding varied between 22% and 100% (Selzner et al, 2001). Better results in Japanese series were attributed to the fact that most patients with cirrhosis in Japanese series were nonalcoholic. Although alcohol-related chronic liver disease has been considered by some authors to be at higher risk for postoperative complications because of other coexisting diseases and malnutrition, others found no significant difference in the outcomes of patients with alcoholic versus nonalcoholic cirrhosis (Rikkers et al, 1998; Selzner et al, 2001; Sugiura & Futagawa, 1973). In patients with cirrhosis, the major cause of mortality is hepatic decompensation and not variceal bleeding. However, in patients with a noncirrhotic etiology, variceal bleeding rather than liver failure is the common cause of death following acute variceal bleeding. In an analysis of 3588 operated patients from 59 Japanese centers, Inokuchi (1985) reported that postoperative variceal bleeding accounted for only 8. The high rate of mortality is reported in Child-Pugh C cirrhosis who are taken up for surgery for acute variceal bleeding which was not controlled by other means, as a "last ditch effort" (Liu, 2014). Qazi and colleagues (2006) reported mortality in Child-Pugh A, B, and C as 12% to 14%, 30% to 50%, and 80%, respectively, for the modified Sugiura procedure. Operative Morbidity the outcomes, including morbidity and mortality, in various devascularization operations are summarized in Tables 84. The morbidity is related to the underlying liver dysfunction and the surgical technique used. The rate of esophageal leakage and stenosis with the Sugiura and modified Sugiura procedures incorporating esophageal transection are 5% to 14% and 2% to 28%, respectively. Some authors have suggested that avoiding esophageal transection can avoid these complications while maintaining comparable rebleed rates (Johnson et al, 2006; Zhang et al, 2014). However, others have argued that esophageal transection is a very important component of the procedure to reduce variceal recurrence and will result in a significantly higher failure rate if esophageal transection is not performed (Sugiura & Futagawa, 1984). We routinely perform esophageal transection and avoid this only in cases of a friable esophagus or one that has undergone recent multiple sessions of sclerotherapy. The incidence of postoperative ascites has ranged widely, from 3% to 33%, based on the severity of cirrhosis, extent of splanchnic venous thrombosis, and preoperative presence of ascites. In series reporting a high rate of postoperative ascites, it usually resolves within a few months (Selzner et al, 2001). Devascularization procedures do not usually result in deterioration of liver function, and encephalopathy is generally not exacerbated. Although patients with Child-Pugh C are likely to have poor outcomes with high morbidity regardless of the type of nontransplant treatment modality, devascularization cannot be considered to be contraindicated because it is the only salvage option that remains when other modalities have either failed or are not available (Qazi et al, 2006) (see Chapter 77). Portal vein thrombosis may be linked to thrombocythemia or to a decrease in portal blood flow after splenectomy (Han et al 2014; Takenaka et al, 1990; Zhang et al, 2012; Zheng et al, 2013) and affects 5. The actual rate may be higher because reported rates usually include only symptomatic cases. Unexplained fever and abdominal pain by the end of the first postoperative week should be assessed by ultrasound Doppler and contrast computed tomography. Acute portal vein thrombosis should be treated with anticoagulation for a period of 3 to 6 months with careful monitoring. Efficacy: Control of Variceal Hemorrhage Devascularization procedures are effective in controlling variceal bleeding. They also address the underlying hypersplenism as splenectomy is a part of the procedure. Overall, devascularization procedures have a rebleeding rate of 5% to 16% and mortality rate of 1% to 7%, without risk of encephalopathy (Battaglia et al, 1996; Coelho et al 2014; Liu et al, 2013; Hayashi et al; 2013; Mercado et al, 1999; Raia et al, 1994). The best way to evaluate a devascularization procedure would be determining its efficacy in controlling acute bleeding and prevention of rebleeding. The beneficial effect will translate into overall improved survival of these patients, dependent on the underlying etiology and liver functional status. Immediate control of bleeding is achieved in almost all cases: 95% to 100% (Sharma et al, 2007; Hassab, 1967; Inokuchi, 1985; Liu, 2013; Mathur et al, 1997; Sugiura & Futagawa, 1984). Although the initial procedure, such as esophageal transection alone or splenectomy alone, does not yield satisfactory results, the Hassab procedure, the Sugiura procedure, and its modifications resulted in good control of variceal bleeding, with a resultant decrease in recurrence of bleeding and improved survival. The reported benefits of the Hassab and Sugiura procedure and its modifications include low mortality, a low incidence of recurrent bleeding, and absence of encephalopathy when compared with published series of nonselective shunts. Both procedures report excellent control of acute bleeding in emergency cases and regression of varices in 91% and 97% patients, respectively, with the devascularization procedures described by these authors (Hassab, 1967; Sugiura & Futagawa, 1984).
The opsonic activity correlates with ascites protein levels depression angle definition generic 25 mg clozapine otc, and patients with ascites protein less than 10 g/L have a greater probability of developing peritonitis than do those with higher ascites protein levels (Llach et al depression criteria 25 mg clozapine amex, 1992; Runyon mood disorder not otherwise specified purchase clozapine mastercard, 1986) natal depression definition 100 mg clozapine order with visa. Obvious symptoms include fever and abdominal pain depression symptoms toddlers discount clozapine 50 mg without prescription, but more subtle symptoms are those of decompensation of liver disease, including acute renal dysfunction, encephalopathy, and jaundice. Bacterascites is the term applied to a positive ascites culture in the absence of neutrascites. Limited information on this subset of patients suggests that the infection may be transient (Pelletier et al, 1991). If the ascites is bloody or the paracentesis traumatic, the neutrophil counts in the fluid should be adjusted for the number of red blood cells present. Inoculating ascites directly into blood culture media at the bedside increases the likelihood that microbiologic organisms will be recovered (Runyon et al, 1990). It has been estimated that for every five patients whose ascites is immediately inoculated into culture media rather than delayed by inoculation in the microbiology laboratory, one additional bacterial infection will be detected. Particularly high neutrophil counts or the recovery of multiple microbiologic organisms should raise concern for secondary peritonitis that would reflect either a suppurative abdominal infection or bowel perforation. In common practice, the antibiotics of choice are third-generation cephalosporins. Five days of therapy are as efficacious as 10 days of therapy (Runyon et al, 1991). Subgroup analysis suggests that patients with preexisting renal impairment or a serum bilirubin level greater than 4 mg/dL are most likely to benefit from this intervention. Hepatic Cirrhosis, Portal Hypertension, and Hepatic Failure Chapter 81 Management of ascites in cirrhosis and portal hypertension 1195. Andreu M, et al: Risk factors for spontaneous bacterial peritonitis in cirrhotic patients with ascites, Gastroenterology 104:11331138, 1993. Arroyo V, et al: Refractory ascites: definition and mechanism, Minerva Med 78:675680, 1987. Arroyo V, et al: Plasma renin activity and urinary sodium excretion as prognostic indicators in nonazotemic cirrhosis with ascites, Ann Intern Med 94:198201, 1981. Arroyo V, et al: Antidiuretic hormone and the pathogenesis of water retention in cirrhosis with ascites, Semin Liver Dis 14:4458, 1994. Arroyo V, et al: Sympathetic nervous activity, renin-angiotensin system and renal excretion of prostaglandin E2 in cirrhosis. Relationship to functional renal failure and sodium and water excretion, Eur J Clin Invest 13:271278, 1983. Bauer M, et al: Functional significance of endothelin B receptors in mediating sinusoidal and extrasinusoidal effects of endothelins in the intact rat liver, Hepatology 31:937947, 2000. Benet A, et al: Diagnosis of hepatic hydrothorax in the absence of ascites by intraperitoneal injection of 99m-Tc-Fluor colloid, Postgrad Med J 68:153, 1992. Bernardi M, et al: Aldosterone related blood volume expansion in cirrhosis before and during the early phase of ascites formation, Gut 24:761766, 1983. Blendis L, Wong F: Intravenous albumin with diuretics: protean lessons to be learnt Coulon S, et al: Angiogenesis in chronic liver disease and its complications, Liver Int 31:146162, 2011. Dhanasekaran R, et al: Transjugular intrahepatic portosystemic shunt for symptomatic refractory hepatic hydrothorax in patients with cirrhosis, Am J Gastroenterol 105:635641, 2010. Facciorusso A, et al: Role of vaptans in the management of hydroelectrolytic imbalance in liver cirrhosis, World J Hepatol 6:793799, 2014. Garcia-Martinez R, et al: Albumin infusion improves renal blood flow autoregulation in patients with acute decompensation of cirrhosis and acute kidney injury, Liver Int 35:335343, 2015. Gentilini P, et al: Ascites and hepatorenal syndrome during cirrhosis: two entities or the continuation of the same complication Gilani N, et al: the safety and feasibility of large volume paracentesis performed by an experienced nurse practitioner, Ann Hepatol 8:359 363, 2009. Gines A, et al: Randomized trial comparing albumin, dextran 70, and polygeline in cirrhotic patients with ascites treated by paracentesis, Gastroenterology 111:10021010, 1996. Gines P, Guevara M: Hyponatremia in cirrhosis: pathogenesis, clinical significance, and management, Hepatology 48:10021010, 2008. Gines P, et al: Comparison of paracentesis and diuretics in the treatment of cirrhotics with tense ascites. Gines P, et al: Compensated cirrhosis: natural history and prognostic factors, Hepatology 7:122128, 1987b. Gines P, et al: Transjugular intrahepatic portosystemic shunting versus paracentesis plus albumin for refractory ascites in cirrhosis, Gastroenterology 123:18391847, 2002. Gines P, et al: Management of cirrhosis and ascites, N Engl J Med 350:16461654, 2004. Gines P, et al: Effects of satavaptan, a selective vasopressin V(2) receptor antagonist, on ascites and serum sodium in cirrhosis with hyponatremia: a randomized trial, Hepatology 48:204213, 2008. Harris K, Chalhoub M: the use of a PleurX catheter in the management of recurrent benign pleural effusion: a concise review, Heart Lung Circ 21:661665, 2012. Hennenberg M, et al: Mechanisms of extrahepatic vasodilation in portal hypertension, Gut 57:13001314, 2008. Huang P-M, et al: the morphology of diaphragmatic defects in hepatic hydrothorax: thoracoscopic finding, J Thorac Cardiovasc Surg 130:141145, 2005. Iwakiri Y, et al: Vascular biology and pathobiology of the liver: Report of a single-topic symposium, Hepatology 47:17541763, 2008. Llach J, et al: Incidence and predictive factors of first episode of spontaneous bacterial peritonitis in cirrhosis with ascites: relevance of ascitic fluid protein concentration, Hepatology 16:724727, 1992. Malinchoc M, et al: A model to predict poor survival in patients undergoing transjugular intrahepatic portosystemic shunts, Hepatology 31:864871, 2000. Mouroux J, et al: Management of pleural effusion of cirrhotic origin, Chest 109:10931096, 1996. Pelletier G, et al: Asymptomatic bacterascites: is it spontaneous bacterial peritonitis Relationship between the diuretic response and the activity of the renin-aldosterone system, Gastroenterology 84:961968, 1983. Rossle M, et al: A comparison of paracentesis and transjugular intrahepatic portosystemic shunting in patients with ascites, N Engl J Med 342:17011707, 2000. Roussos A, et al: Hepatic hydrothorax: pathophysiology diagnosis and management, J Gastroenterol Hepatol 22:13881393, 2007. Ruiz-Del-Arbol L, et al: Paracentesis-induced circulatory dysfunction: mechanism and effect on hepatic hemodynamics in cirrhosis, Gastroenterology 113:579586, 1997. A randomized controlled study of 100 patients, Gastroenterology 100:17371742, 1991. Salerno F, et al: Transjugular intrahepatic portosystemic shunt for refractory ascites: a meta-analysis of individual patient data, Gastroenterology 133:825834, 2007. Shah V, et al: Liver sinusoidal endothelial cells are responsible for nitric oxide modulation of resistance in the hepatic sinusoids, J Clin Invest 100:29232930, 1997. Sola-Vera J, et al: Randomized trial comparing albumin and saline in the prevention of paracentesis-induced circulatory dysfunction in cirrhotic patients with ascites, Hepatology 37:11471153, 2003. Sort P, et al: Effect of intravenous albumin on renal impairment and mortality in patients with cirrhosis and spontaneous bacterial peritonitis, N Engl J Med 341:403409, 1999. Tito L, et al: Recurrence of spontaneous bacterial peritonitis in cirrhosis: frequency and predictive factors, Hepatology 8:2731, 1988. Yutaka Y, et al: A novel surgical approach to refractory hepatic hydrothorax, Ann Thorac Surg 96:e75e76, 2013. Trotter Variceal bleeding is a major life-threatening complication of cirrhosis associated with a high mortality of up to 50% within 30 days of presentation. However, while medical management of this condition has improved, so have patient outcomes (Carbonell, 2004; El-Serag, 2000; Jairath, 2014; Jamal, 2008a, 2008b; Stokkeland, 2006). Specifically, the 30-day mortality has decreased from 30% to 43% in earlier eras to only 15% to 21% currently. Likewise, the in-hospital mortality has declined to just greater than 10% (Jamal, 2008a). These lower mortality rates may be attributed to the combined effects of more aggressive clinical treatment protocols and the development of more effective therapies, such as esophageal band ligation; intravenous vasoactive medications, such as octreotide and terlipressin; and antibiotic prophylaxis. At this time, nearly all patients admitted for variceal bleeding receive antibiotic prophylaxis (94%) and endoscopic therapy (90%) within 24 hours compared with 2% and 6%, respectively, 20 years ago (Carbonell, 2004). In addition, evidence suggests that prophylactic therapies have reduced the overall rate of bleeding; currently, 15% fewer patients are hospitalized than a decade ago (Jamal, 2008b). This finding underscores the clinical utility of early diagnosis of esophageal varices through aggressive screening strategies, careful surveillance for small varices, and prophylactic therapy with -blockers or endoscopic band ligation before or after variceal bleeding occurs. The risk of variceal rupture is dependent upon an increase in wall tension across the varix resulting from elevations in the transmural pressure, increased size of the varix, and decreased wall thickness. Additional risk factors may include the presence of ascites, increased plasma volume, and excessive blood transfusions (Castaneda, 2001). Size of esophageal varices can be categorized either semiquantitatively or quantitatively. Varices can be classified as small (minimal elevation of veins above the mucosa), medium (tortuous veins, occupying less than one third of the lumen), or large (occupying greater than one third of the lumen). An alternative classification based on two sizes, small or large, defines small varices as less than or equal to 5 mm and large varices greater than 5 mm (de Franchis, 1992). However, as recognized by experienced endoscopists, the interobserver agreement in size estimation of esophageal varices can vary considerably (Bendtsen, 1990). The likelihood of discovering varices in a particular patient is largely dependent on the severity of the underlying liver disease. In cirrhotic patients without varices, the risk of developing varices is 5% to 10% per year (Groszmann, 2005; Merli, 2003). The variceal size is important because of its direct relationship to bleeding and need for prophylaxis (Zoli, 1996). Patients with small varices progress to large varices at a rate of approximately 7% per year (Cales, 1999; Merkel, 2004). The overall risk of symptomatic bleeding from esophageal varices is approximately 10% per year. For patients without varices, the yearly bleeding risk is only 1% per year, which 1196 D. Hepatic Cirrhosis, Portal Hypertension, and Hepatic Failure Chapter 82 Medical management of bleeding varices 1197 of variceal hemorrhage or a recent bleeding event, including red spots, red wale markings, and fibrin plugs. As the risk of variceal progression and bleeding depend on both the severity of the underlying liver disease and appearance of the varices at endoscopy, patients with decompensated liver disease and highrisk endoscopic features (large varices, red wale markings) require more aggressive screening protocols. In addition, screening may be appropriate in selected patients with bridging fibrosis because 16% may have esophageal varices (Sanyal, 2006). Once endoscopy has been performed, the presence or absence of varices has been established, and the varices have been graded according to severity, the requirement for further screening, surveillance, and therapy may be determined. In general, treatment recommendations for medium varices are the same as for large varices. In patients without varices, the recommended screening interval to detect the development of varices is within 3 years. As the yearly risk of new varices is approximately 5% to 10% in this population, the cumulative risk of developing of varices over 3 years would be only approximately 25%, and during this interval, the risk of bleeding is quite small (1% per year) (Groszmann, 2005; Merli, 2003). Prophylactic therapy with -blockers to prevent the development of varices or bleeding in cirrhotic patients without varices is not effective, as determined by two randomized studies, one with propranolol (Cales, 1999) and one with timolol (Groszmann, 2005). Because both studies failed to show benefit with nonselective -blockers in these populations, pharmacologic prevention of varices, or "preprimary prophylaxis," is not possible at present. In a patient with small varices identified at initial endoscopy, the yearly risk of developing large varices is approximately 7% (Cales, 1999; Merkel, 2004), and the risk of hemorrhage is only 5%. Repeat endoscopy should be performed at 2 years or yearly in the case of decompensation. Prophylactic treatment with -blockers in patients with small varices has led to mixed results. In these studies, one trial reported a higher 2 year rate of developing large varices (31%) with treatment compared with placebo (14%); however, the dropout rate in this study was very high. Another trial reported a lower rate of progression to large varices with -blockers (11%) versus placebo (37%) after 3 years. The risk of bleeding was lower (12%) at 5 years with treatment compared with placebo (22%). However, the benefit of -blockers was mitigated by the fact that the rate of bleeding in the placebo group, which was started on -blockers once large varices were discovered, was the same as the treatment group once -blocker therapy was initiated. In addition, the rate of withdrawal because of side effects was higher with -blockers (10%) compared with placebo (1%). Because of these mixed results, patients with small varices should receive prophylactic -blocker therapy only if they have risk factors for bleeding, such as advanced liver disease or red wale markings. In patients not receiving -blockers, repeat endoscopy is recommended every other year or annually for decompensation, and repeat endoscopy is not required for patients taking -blockers. In patients with medium or large varices identified at screening endoscopy, prophylactic therapy is clearly recommended, as described below. In addition, endoscopic sclerotherapy has no role in prophylaxis (see Chapter 83). Noninvasive screening techniques have several potential advantages versus endoscopy. They are generally more widely accepted by patients, which could potentially increase adherence to screening protocols. The four general types of alternative screening modalities are (1) blood chemistries, (2) crosssectional imaging, (3) transient elastography, and (4) alternative endoscopic techniques, such as ultrathin endoscopy and capsule endoscopy. Laboratory-Based Markers the least invasive means of screening for varices is analysis of blood chemistries. There has been increasing interest in recent years in evaluating the use of inflammatory markers to predict the presence of esophageal varices. The pathophysiologic rationale is that portal hypertension is directly related to liver injury and fibrosis. The presence and level of specific inflammatory mediators in the serum could indirectly predict liver damage and therefore severity of portal hypertension.
Arakura N depression jokes one liners purchase clozapine line, et al: Efficacy of preoperative endoscopic nasobiliary drainage for hilar cholangiocarcinoma depression feeling overwhelmed buy clozapine no prescription, J Hepatobiliary Pancreat Surg 16(4):473477 bipolar depression for a year hoping for mania purchase clozapine 25 mg with mastercard, 2009 anxiety 9 to 5 purchase on line clozapine. Belghiti J: Arguments for a selective approach of preoperative portal vein embolization before major hepatic resection mood disorder worksheets clozapine 25 mg cheap, J Hepatobiliary Pancreat Surg 11(1):2124, 2004. Ebata T, et al: Pathological appraisal of lines of resection for bile duct carcinoma, Br J Surg 89(10):12601267, 2002. Ebata T, et al: Right hepatopancreatoduodenectomy: improvements over 23 years to attain acceptability, J Hepatobiliary Pancreat Surg 14(2):131135, 2007. Ebata T, et al: Hepatopancreatoduodenectomy for cholangiocarcinoma: a single-center review of 85 consecutive patients, Ann Surg 256(2):297305, 2012a. Ebata T, et al: Portal vein embolization before extended hepatectomy for biliary cancer: current technique and review of 494 consecutive embolizations, Dig Surg 29(1):2329, 2012b. Gertsch P, et al: Multiple tumors of the biliary tract, Am J Surg 159(4):386388, 1990. Higuchi R, et al: Prognostic relevance of ductal margins in operative resection of bile duct cancer, Surgery 148(1):714, 2010. Hwang S, et al: Sequential preoperative ipsilateral hepatic vein embolization after portal vein embolization to induce further liver regeneration in patients with hepatobiliary malignancy, Ann Surg 249(4): 608616, 2009. Igami T, et al: Clinicopathologic study of cholangiocarcinoma with superficial spread, Ann Surg 249(2):296302, 2009a. Itoh S, et al: Assessment of the pancreatic and intrapancreatic bile ducts using 0. Jonas S, et al: Radical surgery for hilar cholangiocarcinoma, Eur J Surg Oncol 34(3):263271, 2008. Kamiya S, et al: the value of bile replacement during external biliary drainage: an analysis of intestinal permeability, integrity, and microflora, Ann Surg 239(4):510517, 2004. Kanai M, et al: Preoperative intrahepatic segmental cholangitis in patients with advanced carcinoma involving the hepatic hilus, Surgery 119(5):498504, 1996. Kawai K, et al: Inchinkoto, an herbal medicine, exerts benefical effects in the rat liver under stress with hepatic ischemia-reperfusion and subsequent hepatectomy, Ann Surg 251(4):692700, 2010. Kawakami H, et al: Preoperative biliary drainage for hilar cholangiocarcinoma: which stent should be selected Kawakami H, et al: Endoscopic nasobiliary drainage is the most suitable preoperative biliary drainage method in the management of patients with hilar cholangiocarcinoma, J Gastroenterol 46(2):242 248, 2011b. Kawasaki S, et al: Radical operation after portal embolization for tumor of hilar bile duct, J Am Coll Surg 178(5):480486, 1994. Kawashima H, et al: Preoperative endoscopic nasobiliary drainage in 164 consecutive patients with suspected perihilar cholangiocarcinoma: a retrospective study of efficacy and risk factors related to complications, Ann Surg 257(1):121127, 2013. Kenjo A, et al: Risk stratification of 7,732 hepatectomy cases in 2011 from the National Clinical Database for Japan, J Am Coll Surg 218(3):412422, 2014. Kinoshita H, et al: Preoperative portal vein embolization for hepatocellular carcinoma, World J Surg 10(5):803808, 1986. Klatskin G: Adenocarcinoma of the hepatic duct at its bifurcation within the porta hepatis. Konishi M, et al: Clinical impact of intraoperative histological examination of the ductal resection margin in extrahepatic cholangiocarcinoma, Br J Surg 97(9):13631368, 2010. Kozuka S, et al: Evolution of carcinoma in the extrahepatic bile ducts, Cancer 54(1):6572, 1984. Kurosaki I, et al: Synchronous primary tumors of the extrahepatic bile duct and gallbladder, J Surg Oncol 65(4):258262, 1997. Kyokane T, et al: An experimental study of selective intrahepatic biliary ablation with ethanol, J Surg Res 96(2):188196, 2001. Maguchi H, et al: Preoperative biliary drainage for hilar cholangiocarcinoma, J Hepatobiliary Pancreat Surg 14(5):441446, 2007. Makuuchi M, et al: Preoperative portal embolization to increase safety of major hepatectomy for hilar bile duct carcinoma: a preliminary report, Surgery 107(5):521527, 1990. Matsumoto N, et al: Role of anatomical right hepatic trisectionectomy for perihilar cholangiocarcinoma, Br J Surg 101(3):261268, 2014. Miura S, et al: Bismuth classification is associated with the requirement for multiple biliary drainage in preoperative patients with malignant perihilar biliary stricture, Surg Endosc 29(7):18621870, 2015. Mizutani T, et al: Does inchinkoto, a herbal medicine, have hepatoprotective effects in major hepatectomy Nagino M, et al: Percutaneous transhepatic biliary drainage in patients with malignant biliary obstruction of the hepatic confluence, Hepatogastroenterology 39(4):296300, 1992. Nagino M, et al: Right or left trisegment portal vein embolization before hepatic trisegmentectomy for hilar bile duct carcinoma, Surgery 117(6):677681, 1995. Nagino M, et al: Selective percutaneous transhepatic embolization of the portal vein in preparation for extensive liver resection: the ipsilateral approach, Radiology 200(2):559563, 1996. Nagino M, et al: Right trisegment portal vein embolization for biliary tract carcinoma: technique and clinical utility, Surgery 127(2):155 160, 2000a. Nagino M, et al: Portal and arterial embolization before extensive liver resection in patients with markedly poor functional reserve, J Vasc Interv Radiol 11(8):10631068, 2000b. Nagino M, et al: Two hundred forty consecutive portal vein embolizations before extended hepatectomy for biliary cancer: surgical outcome and long-term follow-up, Ann Surg 243(3):364372, 2006. Nagino M, et al: Preoperative biliary drainage for biliary tract and ampullary carcinomas, J Hepatobiliary Pancreat Surg 15(1):2530, 2008. Nagino M, et al: Evolution of surgical treatment for perihilar cholangiocarcinoma: a single-center 34-year review of 574 consecutive resections, Ann Surg 2012. Nakanishi Y, et al: Extrahepatic bile duct carcinoma with extensive intraepithelial spread: a clinicopathological study of 21 cases, Mod Pathol 21(7):807816, 2008. Nakanishi Y, et al: Impact of residual in situ carcinoma on postoperative survival in 125 patients with extrahepatic bile duct carcinoma, J Hepatobiliary Pancreat Surg 2009. Nakeeb A, et al: Cholangiocarcinoma: a spectrum of intrahepatic, perihilar, and distal tumors, Ann Surg 224(4):463473, discussion 473465, 1996. Natsume S, et al: Hepatopancreatoduodenectomy for anastomotic recurrence from residual cholangiocarcinoma: report of a case, Surg Today 4(5):952956, 2014. Nimura Y: Staging of biliary carcinoma: cholangiography and cholangioscopy, Endoscopy 25(1):7680, 1993. Nimura Y, et al: Cholangioscopic differentiation of biliary strictures and polyps, Endoscopy 21(Suppl 1):351356, 1989. Nimura Y, et al: Technique of inserting multiple biliary drains and management, Hepatogastroenterology 42(4):323331, 1995. Nimura Y, et al: Aggressive surgical treatment of hilar cholangiocarcinoma, J Hepatobiliary Pancreat Surg 5(1):5261, 1998. Nishio H, et al: Most informative projection for portography: quantitative analysis of 47 percutaneous transhepatic portograms, World J Surg 27(4):433436, 2003. Nomura T, et al: Cholangitis after endoscopic biliary drainage for hilar lesions, Hepatogastroenterology 44(17):12671270, 1997. Nomura T, et al: Bacteribilia and cholangitis after percutaneous transhepatic biliary drainage for malignant biliary obstruction, Dig Dis Sci 44(3):542546, 1999. Ogata Y, et al: Role of bile in intestinal barrier function and its inhibitory effect on bacterial translocation in obstructive jaundice in rats, J Surg Res 115(1):1823, 2003. Ohkubo M, et al: Surgical anatomy of the bile ducts at the hepatic hilum as applied to living donor liver transplantation, Ann Surg 239(1):8286, 2004. Radtke A, et al: Computer-assisted surgery planning for complex liver resections: when is it helpful Rayes N, et al: Early enteral supply of fiber and lactobacilli versus conventional nutrition: a controlled trial in patients with major abdominal surgery, Nutrition 18(78):609615, 2002a. Rayes N, et al: Early enteral supply of lactobacillus and fiber versus selective bowel decontamination: a controlled trial in liver transplant recipients, Transplantation 74(1):123127, 2002b. Saiki S, et al: Preoperative internal biliary drainage is superior to external biliary drainage in liver regeneration and function after hepatectomy in obstructive jaundiced rats, Ann Surg 230(5):655 662, 1999. Sakamoto E, et al: the pattern of infiltration at the proximal border of hilar bile duct carcinoma: a histologic analysis of 62 resected cases, Ann Surg 227(3):405411, 1998. Sasaki R, et al: Significance of ductal margin status in patients undergoing surgical resection for extrahepatic cholangiocarcinoma, World J Surg 31(9):17881796, 2007. Shigeta H, et al: Bacteremia after hepatectomy: an analysis of a singlecenter, 10-year experience with 407 patients, Langenbecks Arch Surg 387(34):117124, 2002. Shingu Y, et al: Clinical value of additional resection of a marginpositive proximal bile duct in hilar cholangiocarcinoma, Surgery 147(1):4956, 2010. Sugawara G, et al: Perioperative synbiotic treatment to prevent postoperative infectious complications in biliary cancer surgery: a randomized controlled trial, Ann Surg 244(5):706714, 2006. Sugawara G, et al: the effect of preoperative biliary drainage on infectious complications after hepatobiliary resection with cholangiojejunostomy, Surgery 153(2):200210, 2013. Suzuki H, et al: Internal biliary drainage, unlike external drainage, does not suppress the regeneration of cholestatic rat liver after partial hepatectomy, Hepatology 20(5):13181322, 1994. Taguchi Y, et al: the determination of bile leakage in complex hepatectomy based on the guidelines of the International Study Group of Liver Surgery, World J Surg 38(1):168176, 2014. Takahashi Y, et al: Percutaneous transhepatic biliary drainage catheter tract recurrence in cholangiocarcinoma, Br J Surg 97(12):1860 1866, 2010. Takamoto T, et al: Planning of anatomical liver segmentectomy and subsegmentectomy with 3-dimensional simulation software, Am J Surg 206(4):530538, 2013. Takayasu K, et al: Intrahepatic portal vein branches studied by percutaneous transhepatic portography, Radiology 154(1):3136, 1985. Wakai T, et al: Impact of ductal resection margin status on long-term survival in patients undergoing resection for extrahepatic cholangiocarcinoma, Cancer 103(6):12101216, 2005. Watanabe S, et al: Choleretic effect of inchinkoto, a herbal medicine, on livers of patients with biliary obstruction due to bile duct carcinoma, Hepatol Res 39(3):247255, 2009. Although frequently seen with hilar cholangiocarcinoma (see Chapters 51B and 103C), hilar obstruction (Chapter 42), and even intraductal tumor, can result from other common malignancies, such as breast, pancreatic, and colorectal cancers (Chapters 62, 92, 93, and 94). Significant technical progress has occurred in both endoscopic (see Chapter 29) and percutaneous biliary drainage (Chapters 13 and 30), allowing for safer palliative treatment of patients with such obstructions. Because these patients may be asymptomatic at presentation, the goals of treatment should be clearly defined before the physician commits the patient to even a minimally invasive procedure. With the exception of patients who are clearly in a palliative situation, these patients are ideally discussed in a multidisciplinary group with hepatobiliary surgeons, interventional radiologists, oncologists, and gastroenterologists to outline a plan of treatment. Accepted indications for palliative biliary drainage in these patients include intractable pruritus, cholangitis, the need to restore liver function to allow for administration of chemotherapeutic agents with biliary metabolism/excretion, access for intraluminal brachytherapy or choledochoscopy, and diversion for bile leak. Many physicians have the impression that patients feel better and have improved performance status after relief of jaundice, but this has never been confirmed in clinical studies. Controversy remains with regard to the role of biliary drainage before surgery (Johnson & Ahrendt, 2006; Mezhir et al, 2009; Wang et al, 2008). Although preoperative drainage of the remnant liver is often performed for patients who are surgical candidates but deeply jaundiced at presentation, this practice remains controversial (Fang et al, 2012) (see Chapters 51A and 103C). When preoperative drainage is necessary for relief of symptoms (pruritus, cholangitis), when neoadjuvant therapy is planned or when surgery will be delayed, endoscopic methods are preferred for low bile duct obstruction because of the lower complication rate, whereas high obstruction is treated with carefully targeted percutaneous methods to reduce the risk of cholangitis in undrained segments (Saxena et al, 2015). Patients with high bile duct obstruction, particularly when the obstruction extends above the hilus, have traditionally been treated percutaneously. The success rate of percutaneous drainage is higher, and the complication rate lower, when compared with endoscopic methods (Leng et al, 2014; Rerknimitr et al, 2004). This perspective is evolving while endoscope technology becomes more advanced and while endoscopists become better trained and more experienced in wire-guided procedures, with access to better guidewires and stents. Currently, however, high bile duct obstruction, with rare exception, should be drained percutaneously. The two major drawbacks to endoscopic drainage for high bile duct obstruction have been the lack of ability to target reliably a specific area of the liver for drainage and the risk of contamination, by retrograde injection of contrast, of parts of the biliary tree that will not be drained. Pruritus, cholangitis, and the need to lower the bilirubin to administer certain chemotherapeutic agents, on the other hand, are all accepted indications for biliary drainage. Postoperative bile leaks that require drainage for diversion may develop in patients who have undergone biliary-enteric bypass as part of curative resection for a benign or malignant lesion. In some cases, access to the biliary tree may be undertaken as a method of delivering local treatment 846 C. A realistic idea of what is feasible endoscopically and percutaneously is important when the initial decision is made regarding who will treat the patient. This depends on a thorough understanding of the goal of treatment as well as knowledge of the skill level of the interventional radiologists and endoscopists available to care for the patient. Ultrasound is often used to establish the presence of dilated bile ducts, identify the level of obstruction, evaluate portal vein patency, and demonstrate intraductal tumor, but ultrasound is not adequate for drainage planning. In addition, if the patient is unable to breath-hold during image acquisition, the resulting study will almost certainly be suboptimal. All this information is critical to make prognostic assessments and determine the best approach for drainage. It is important to determine the amount of functional parenchyma that can be drained. Functional parenchyma is the part of the liver that is not replaced by tumor and that has an intact portal venous supply. If 75% of the hepatic parenchyma is replaced by tumor, even if it were possible to drain the entire biliary tree, it is unlikely that normal hepatic function or normalization of serum bilirubin level would result. The portal vein provides the trophic blood supply to the liver, and occlusion results in atrophy of the affected segment(s), particularly when the ipsilateral bile duct is also occluded (see Chapter 5) (Hann et al, 1996). When portal vein occlusion causes atrophy, drainage of the atrophied liver does not improve liver function. In addition, it is frequently difficult to cross out of the obstructed/atrophied portion of the liver. Central tumors frequently obstruct the right and the left biliary tree, isolating them from one another. Despite this, endoscopic management may be indicated in some cases of high bile duct obstruction. Development of the linear-array echoendoscope has allowed endoscopic ultrasound-guided biliary drainage procedures, including choledochoduodenostomy and hepaticogastrostomy (Giovannini et al, 2012).
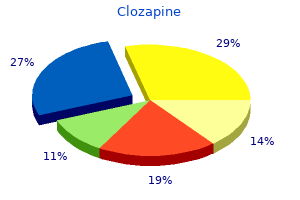
Cirenei A mood disorder 2 clozapine 50 mg otc, Bertoldi I: Evolution of surgery for liver hydatidosis from 1950 to today: analysis of a personal experience anxiety service dog buy generic clozapine 100 mg online, World J Surg 25:87 92 severe depression jesus clozapine 50 mg buy low cost, 2001 anxiety 8 year old son order clozapine with a visa. Daradkeh S bipolar depression evaluation order clozapine 25 mg fast delivery, et al: Predictors of morbidity and mortality in the surgical management of hydatid cyst of the liver, Langenbecks Arch Surg Dtsch Ges Für Chir 392:3539, 2007. Dervenis C, et al: Changing concepts in the management of liver hydatid disease, J Gastrointest Surg 9:869877, 2005. De Werra C, et al: Hydatid disease of the liver: thirty years of surgical experience, Chir Ital 59:611625, 2007. Diez Valladares L, et al: Hydatid liver cyst perforation into the digestive tract, Hepatogastroenterology 45:21102114, 1998. Doiz O, et al: Western blot applied to the diagnosis and post-treatment monitoring of human hydatidosis, Diagn Microbiol Infect Dis 41(3): 139142, 2001. Dziri C, et al: Management of cystic echinococcosis complications and dissemination: where is the evidence Eckert J, Deplazes P: Biological, epidemiological, and clinical aspects of echinococcosis, a zoonosis of increasing concern, Clin Microbiol Rev 17:107135, 2004. Eckert J, et al, editors: World Health Organization/International Office of Epizootics Manual on Echinococcosis in Humans and Animals: a public health problem of global concern, Paris, 2001, World Health Organization/World Organization for Animal Health. Eickhoff A, et al: Endoscopic stenting for postoperative biliary strictures due to hepatic hydatid disease: effectiveness and long-term outcome, J Clin Gastroenterol 37:7478, 2003. Ertem M, et al: Laparoscopically treated liver hydatid cysts, Arch Surg 137(10):11701173, 2002. Filice C, et al: Treatment of echinococcal abdominal cysts, Ultrasound Q 15:223233, 1999. Frider B, et al: Long-term outcome of asymptomatic liver hydatidosis, J Hepatol 30:228231, 1999. Gerazounis M, et al: Bronchobiliary fistulae due to echinococcosis, Eur J Cardio-Thorac Surg 22:306308, 2002. Gourgiotis S, et al: Surgical techniques and treatment for hepatic hydatid cysts, Surg Today 37:389395, 2007. Hegglin D, Deplazes P: Control of Echinococcus multilocularis: strategies, feasibility and cost-benefit analyses, Int J Parasitol 43:327337, 2013. Jerraya H, et al: Predictive factors of recurrence after surgical treatment for liver hydatid cyst, Surg Endosc 29:8693, 2015. Junghanss T, et al: Clinical management of cystic echinococcosis: state of the art, problems, and perspectives, Am J Trop Med Hyg 79:301 311, 2008. Kalovidouris A, et al: Extracapsular (satellite) hydatid cysts, Gastrointest Radiol 17:353356, 1992. Kantarçeken B, et al: Splenic hydatic cyst as a cause of sinistral portal hypertension and isolated gastric variceal bleeding, Turk J Gastroenterol 21:317320, 2010. Kapan M, et al: Postoperative recurrence in hepatic hydatid disease, J Gastrointest Surg 10:734739, 2006. Katkhouda N, et al: Laser resection of a liver hydatid cyst under videolaparoscopy, Br J Surg 79:560561, 1992. Kayaalp C, et al: Biliary complications after hydatid liver surgery: incidence and risk factors, J Gastrointest Surg 6:706712, 2002. Kayaalp C, et al: Distribution of hydatid cysts into the liver with reference to cystobiliary communications and cavity-related complications, Am J Surg 185:175179, 2003a. Kayaalp C, et al: Distribution of hydatid cysts into the liver with reference to cystobiliary communications and cavity-related complications, Am J Surg 185:175179, 2003b. Kern P, et al: European Echinococcosis Registry: human alveolar echinococcosis, Europe, 19822000, Emerg Infect Dis 9:343349, 2003. Keshmiri M, et al: Albendazole versus placebo in treatment of echinococcosis, Trans R Soc Trop Med Hyg 95:190194, 2001. Khoury G, et al: Laparoscopic treatment of hydatid cysts of the liver and spleen, Surg Endosc 14(3):243245, 2000. Kilani T, et al: Hydatid disease of the liver with thoracic involvement, World J Surg 25:4045, 2001. Loinaz C, et al: Long-term biliary complications after liver surgery leading to liver transplantation, World J Surg 25:12601263, 2001. Lv H, et al: Subadventitial cystectomy in the management of biliary fistula with liver hydatid disease, Acta Trop 141:223228, 2015. Manouras A, et al: Endoscopic management of a relapsing hepatic hydatid cyst with intrabiliary rupture: a case report and review of the literature, Can J Gastroenterol 21:249253, 2007. Manterola C, et al: Preoperative albendazole and scolices viability in patients with hepatic echinococcosis, World J Surg 29:750753, 2005. Manterola C, et al: Risk factors of postoperative morbidity in patients with uncomplicated liver hydatid cyst, Int J Surg Lond Engl 12:695 699, 2014. Menezes da Silva A: Hydatid cyst of the liver-criteria for the selection of appropriate treatment, Acta Trop 85:237242, 2003. Nardo B, et al: Radical surgical treatment of recurrent hepatic hydatidosis, Hepatogastroenterology 50:14781481, 2003. Ozaslan E, Bayraktar Y: Endoscopic therapy in the management of hepatobiliary hydatid disease, J Clin Gastroenterol 35:160174, 2002. Ozturk G, et al: Posttraumatic free intraperitoneal rupture of liver cystic echinococcosis: a case series and review of literature, Am J Surg 194:313316, 2007. Palanivelu C, et al: Palanivelu hydatid system for safe and efficacious laparoscopic management of hepatic hydatid disease, Surg Endosc 20(12):19091913, 2006. Reuter S, et al: Structured treatment interruption in patients with alveolar echinococcosis, Hepatology (Baltimore) 39:509517, 2004. Romig T: Epidemiology of echinococcosis, Langenbecks Arch Surg 388(4):209-217, 2003. Romig T, et al: Cyst growth and spontaneous cure in hydatid disease, Lancet 1:861, 1986. Rooh-ul-Muqim: Laparoscopic treatment of hepatic hydatid cyst, J Coll Physicians Surg Pak 21(8):468471, 2011. Saenz de San Pedro B, et al: Anaphylactic shock by rupture of hydatid hepatic cyst. Sahin E, et al: Single-stage transthoracic approach for right lung and liver hydatid disease, J Thorac Cardiovasc Surg 126(3):769773, 2003. Salam A: Laparoscopic treatment of liver hydatid cysts, Surg Laparosc Endosc 6:1621, 1996. Sakhri J, Ben Ali A: [Hydatid cyst of the liver], J Chir (Paris) 141:381 389, 2004. Sbihi Y, et al: Serologic recognitionof hydatid cyst antigens using different purification methods, Diag Micro Infect Dis 24(4):205211, 1996. Seven R, et al: Laparoscopic treatment of hepatic hydatid cysts, Surgery 128:3640, 2000. Siracusano A, Bruschi F: Cystic echinococcosis: progress and limits in epidemiology and immunodiagnosis, Parassitologia 48:6566, 2006. Sonmez G, et al: Is it possible to differentiate between hydatid and simple cysts in the liver by means of diffusion-weighted magnetic resonance imaging Symeonidis N, et al: Complicated liver echinococcosis: 30 years of experience from an endemic area, Scand J Surg 102:171177, 2013. Tagliacozzo S, et al: Surgical treatment of hydatid disease of the liver: 25 years of experience, Am J Surg 201:797804, 2011. Teggi A, et al: Therapy of human hydatid disease with mebendazole and albendazole, Antimicrob Agents Chemother 37:16791684, 1993. Tocchi A, et al: Treatment of hydatid bronchobiliary fistulas: 30 years of experience, Liver Int 27:209214, 2007. Todorov T, et al: Evaluation of response to chemotherapy of human cystic echinococcosis, Br J Radiol 63:523531, 1990. Relative survival and economic analysis in Switzerland over the last 35 years, J Hepatol 49:7277, 2008. Tuxun T, et al: World review of laparoscopic treatment of liver cystic echinococcosis-914 patients, Int J Infect Dis 24:4350, 2014. Vutova K, et al: Effect of mebendazole on human cystic echinococcosis: the role of dosage and treatment duration, Ann Trop Med Parasitol 93:357365, 1999. Man:dog contact and its role in the transmission and control of hydatidosis amongst the Turkana, Ann Trop Med Parasitol 82:343356, 1988. Wen H, et al: Albendazole chemotherapy for human cystic and alveolar echinococcosis in north-western China, Trans R Soc Trop Med Hyg 88:340343, 1994. World Health Organization: Guidelines for treatment of cystic and alveolar echinococcosis in humans. Yilmaz U, et al: Management of postoperative biliary strictures secondary to hepatic hydatid disease by endoscopic stenting, Hepatogastroenterology 45:6569, 1998. Yol S, et al: Open drainage versus overlapping method in the treatment of hepatic hydatid cyst cavities, Int Surg 84:139143, 1999. Yorganci K, Sayek I: Surgical treatment of hydatid cysts of the liver in the era of percutaneous treatment, Am J Surg 184:6369, 2002. Zaharie F, et al: Open or laparoscopic treatment for hydatid disease of the liver Zaouche A, et al: Management of liver hydatid cysts with a large biliocystic fistula: multicenter retrospective study. Zaouche A, et al: Management of liver hydatid cysts with a large biliocystic fistula: multicenter retrospective study, World J Surg 25(1):2839, 2001b. Zhang W, et al: Concepts in immunology and diagnosis of hydatid disease, Clin Microbiol Rev 16(1):1836, 2003. This differentiates these diseases from hydatid cysts (see Chapter 74), cystic hepatobiliary neoplasms (see Chapter 90B), and cystic dilation of the intrahepatic bile ducts (see Chapter 46). Apart from acute complications, the most frequent of which is intracystic bleeding, these symptoms have frequently evolved progressively for a prolonged period of time (years). It may therefore be difficult for clinicians to differentiate those related to the cyst(s) or to the associated conditions. It may even occasionally take some time to understand the true severity of cyst-related symptoms that some patients have unconsciously become used to . When indicated, the aim of treatment in patients with single cysts is to either destroy the epithelial lining of the cyst with sclerotherapy or create a communication between the cyst lumen and the peritoneal cavity by fenestration. Numerous terms have been used to designate this lesion, including biliary cyst, nonparasitic cyst of the liver, benign hepatic cyst, congenital hepatic cyst, unilocular cyst of the liver, and solitary cyst of the liver. The last designation is inappropriate because, as mentioned later, simple cysts are often multiple. Symptoms, when present, occur in larger cysts and are usually associated with intracystic bleeding that changes the morphologic appearance of the cyst. It is in this circumstance that differential diagnosis with other cysts, most notably cystadenomas or hydatid disease, may prove difficult. Pathology and Pathogenesis Macroscopically, a simple cyst of the liver has a spherical or ovoid shape. Large cysts produce atrophy of the adjacent hepatic tissue; a huge cyst may result in complete atrophy of a hepatic lobe, with compensatory hypertrophy of the other lobe. Atrophy respects the large bile ducts and blood vessels, which appear to be abundant in the atrophic tissue in contact with the cyst. Large bile ducts and blood vessels persisting after atrophy may protrude and form folds over the inner surface of the cyst. The cystic fluid is usually clear; however, intracystic bleeding is relatively common, and the fluid may be brownish when this is the case. Stroma is absent in small cysts and reduced to a thin layer of connective tissue in large cysts. An aberrant bile duct would have lost communication with the biliary tree and would dilate progressively. Liver cyst epithelial cells retain differentiated secretory function; they secrete fluid and generate a positive luminal pressure that may be greater than 30 cm H2O, which accounts for most symptoms. The composition of the fluid, which contains water and mineral electrolytes without bile acids and bilirubin, is close to that of the normal secretion of the epithelium of the bile ducts. Based on these observations, the diagnosis of simple cysts should be retained with caution when a large and/or symptomatic cystic mass is observed in a male patient or in a young woman. Because of the high prevalence of simple cysts in adults, the fortuitous coincidence of this lesion and another hepatic or extrahepatic disease is common. The causal relationship between abdominal pain or discomfort and a simple cyst of the liver must be admitted with caution and accepted only if the cyst is large and the other possible causes of the symptoms have been excluded. Liver function tests are normal; abnormal liver function tests in a patient with simple cyst of the liver are not related to the cyst and are caused by another liver disease, except when the cysts compress bile ducts. Ultrasonography is the best procedure for recognizing simple cysts (see Chapter 15). A simple cyst is a circular or oval, totally anechoic lesion, with sharp, smooth borders and strong posterior wall echoes, indicating a well-defined tissue-fluid interface (Liang et al 2005). By 1971, only 350 cases had been reported (Flagg & Robinson, 1967; Moreaux & Bloch, 1971), and early estimates from autopsy studies were less than 1% (Larsen, 1961). The development of imaging techniques revealed, however, that simple cysts are much more frequent. These discrepancies are explained by the usual small size of most cysts, which are discovered only with accurate imaging techniques. An association between simple hepatic cysts and simple renal cysts has been documented (Carrim & Murchison, 2003) but is unexplained. Simple cysts are uncommon before age 40 years, and their incidence increases sharply thereafter. Simple cysts also are larger in adults older than 50 years than in younger individuals (Larsen, 1961).
Clozapine 50 mg order on line. Pediatric Bipolar Disorder.
References
- Felker GM, Lee KL, Bull DA, et al. Diuretic strategies in patients with acute decompensated heart failure. N Engl J Med 2011;364:797.
- Tan EK, Lum SY, Fook-Chong SM, et al. Evaluation of somnolence in Parkinsonis disease: comparison with age- and sex-matched controls. Neurology 2002;58(3):465-8.
- Cathelineau, X., Arroyo, C., Rozel, F. et al. Laparoscopicassisted radical cystectomy: the Montsouris experience after 84 cases. Eur Urol 2005;47:780-784.
- Hofflin JM, Potasman I, Baldwin JC, et al. Infectious complications in heart transplant recipients receiving cyclosporine and corticosteroids. Ann Intern Med. 1987;106:209-216.
