
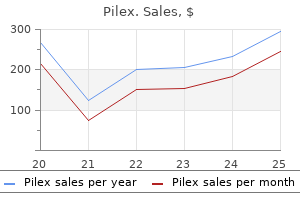
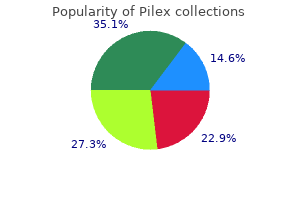
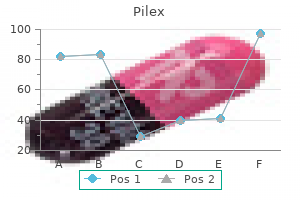
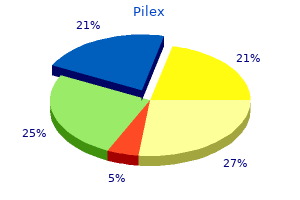
Pilex
| Contato
Página Inicial
Francis D. Ferdinand MD, FRCSEd, FACS, FACC
- Assistant Professor of Surgery, Jefferson Medical College, Philadelphia,
- Pennsylvania
- Associate Investigator, Lankenau Institute for Medical Research
- Division of Thoracic and Cardiovascular Surgery, Lankenau Hospital, Wynnewood,
- Pennsylvania
The mucosa has a urothelium near the bladder man healthcom pay bill pay bill buy cheap pilex 60 caps on-line, pseudostratified epithelium for most of its length mens health diet pdf buy pilex 60 caps without prescription, and finally stratified squamous epithelium near the external urethral orifice mens health recipe book generic pilex 60 caps buy. In males prostate cancer 80 year old discount pilex 60 caps overnight delivery, smooth muscle around the neck of the bladder and nearby urethra is of a different physiological type than the detrusor prostate cancer zone order 60 caps pilex with visa, richly innervated by the sympathetic nervous system and thickened into a collar of muscle called the internal urethral sphincter. Its function seems to be to prevent the reflux of semen into the bladder during ejaculation. In both sexes, however, there is an external urethral sphincter of skeletal muscle encircling the urethra where it passes through the pelvic floor. In the female, it is a tube 3 to 4 cm long bound to the anterior wall of the vagina by fibrous connective tissue. Its opening, the external urethral orifice, lies between the vaginal orifice and clitoris. The male urethra is about 18 cm long and has three regions: (1) the prostatic urethra begins at the urinary bladder and passes for about 2. The male urethra assumes an S shape: It passes downward from the bladder, turns anteriorly as it enters the root of 23. This is ensured by sympathetic pathways that originate in the upper lumbar spinal cord. Postganglionic fibers travel through the hypogastric nerve to the detrusor and the male internal urethral sphincter. The detrusor relaxes to allow the bladder to fill, but the sphincters contract to ensure that urine is retained in the bladder until voluntarily voided. The external urethral sphincter is held closed by somatic motor fibers that travel from the sacral spinal cord via the pudendal nerve to the sphincter. Here, it is controlled by a relatively simple spinal micturition reflex depicted in steps 1 through 4 of figure 23. Filling of the bladder to about 200 mL or more excites stretch receptors in the bladder wall. These issue signals by way of sensory fibers in the pelvic nerve to the sacral spinal cord (usually segments S2S3). Efferent signals from the spinal cord travel back to the bladder muscles by way of motor fibers in the pelvic nerve and a parasympathetic ganglion in the bladder wall. When there is no voluntary control over urination, this reflex alone releases urine from the bladder. This is the case, too, in people with spinal cord injuries that disconnect the brain from the lower spinal cord (see Deeper Insight 23. The inability to consciously control urination is called urinary incontinence, and can result not only from spinal cord injuries but from a number of disease conditions as well as simple aging (see table 23. There are still uncertainties in the scientific community, however, about some of the details. In both sexes, signals from the stretch receptors of the bladder ascend the spinal cord to a nucleus in the pons called the micturition center. Because of this risk, young girls should be taught never to wipe the anus in a forward direction. Cystitis is frequently triggered in women by sexual intercourse ("honeymoon cystitis"). If cystitis is untreated, bacteria can spread up the ureters and cause pyelitis,32 infection of the renal pelvis. Urine stagnation due to renal calculi or prostate enlargement increases the risk of infection. Voluntary control 5 For voluntary control, micturition center in pons receives signals from stretch receptors. Control in the female differs slightly because of the lack of an internal urethral sphincter. Thus, urination can be prompted by fear (via the amygdala) or inhibited by cerebral awareness that the circumstances are inappropriate for urination. Fibers from the micturition center descend the spinal cord through the reticulospinal tracts. Others descend farther to the sacral spinal cord and excite the parasympathetic neurons that relax the internal urethral sphincter and stimulate the detrusor. The initial contraction of the detrusor raises pressure in the bladder, further exciting the stretch receptors that started the process; thus, a positive feedback loop is established that intensifies bladder contraction as urination proceeds. For the bladder to begin emptying, however, one final obstacle must be overcome-the external urethral sphincter. Nerve fibers from the cerebral cortex descend by way of the corticospinal tracts and inhibit sacral somatic motor neurons that normally keep that sphincter constricted. It is this voluntary component of micturition that gives a person conscious control of when to urinate and the ability to stop urination in midstream. Transection of the spinal cord, as in many cervical fractures, disconnects the supraspinal control centers (cerebrum and pons) from the spinal cord circuits that control urination. Bladder control returns as the spinal cord recovers, but is limited to the involuntary micturition reflex. The bladder often cannot empty completely, and there is consequently an increased incidence of cystitis. Males expel the last few milliliters of urine by voluntarily contracting the bulbocavernosus muscle that ensheathes the root of the penis. Results in destruction of glomeruli leading to hematuria, proteinuria, edema, reduced glomerular filtration, and hypertension. Can progress to chronic glomerulonephritis and renal failure, but most individuals recover from acute glomerulonephritis without lasting effect. An abrupt decline in renal function, often due to traumatic damage to the nephrons or a loss of blood flow stemming from hemorrhage or thrombosis. Increase in fluid pressure in the renal pelvis and calyces owing to obstruction of the ureter by kidney stones, nephroptosis, or other causes. Can progress to complete cessation of glomerular filtration and atrophy of nephrons. Occurs in people with too little body fat to hold the kidney in place and in people who subject the kidneys to prolonged vibration, such as truck drivers, equestrians, and motorcyclists. Can twist or kink the ureter, which causes pain, obstructs urine flow, and potentially leads to hydronephrosis. Can result from trauma, drugs, infections, cancer, diabetes mellitus, lupus erythematosus, and other diseases. Loss of plasma protein leads to edema, ascites, hypotension, and susceptibility to infection (because of immunoglobulin loss). Can result from aging; incompetence of the urinary sphincters; bladder irritation; pressure on the bladder in pregnancy; an obstructed urinary outlet so that the bladder is constantly full and dribbles urine (overflow incontinence); uncontrollable urination due to brief surges in bladder pressure, as in laughing or coughing (stress incontinence); and neurological disorders such as spinal cord injuries. In females, the external urethral sphincter may be the only mechanism for urine retention. Retention therefore depends on the strength of the voluntary pelvic floor muscles, which can be compromised by a history of pregnancy or obesity. If the urge to urinate arises at an inconvenient time and one must suppress it, the stretch receptors fatigue and stop firing. As bladder tension increases, however, the signals return with increasing frequency and persistence. Compare and contrast the functions of the internal and external urethral sphincters. In males, the sympathetic nervous system triggers ejaculation and, at the same time, stimulates constriction of the internal urethral sphincter. Some causes of nephron destruction include · hypertension; · chronic or repetitive kidney infections; · trauma from such causes as blows to the lower back or continual vibration from machinery; · prolonged ischemia and hypoxia, as in long-distance runners and swimmers; · poisoning by heavy metals such as mercury and lead and solvents such as carbon tetrachloride, acetone, and paint thinners. These are absorbed into the blood from inhaled fumes or by skin contact and then filtered by the glomeruli. Even when some of the nephrons are irreversibly destroyed, others hypertrophy and compensate for their lost function. When 75% of the nephrons are lost, however, urine output may be as low as 30 mL/h compared with the normal rate of 50 to 60 mL/h. This is insufficient to maintain homeostasis and is accompanied by azotemia and acidosis. Blood is pumped from the radial artery to a dialysis machine (artificial kidney) and returned to the patient by way of a vein. In the dialysis machine, the blood flows through a semipermeable cellophane tube surrounded by dialysis fluid. Urea, potassium, and other solutes that are more concentrated in the blood than in the dialysis fluid diffuse through the membrane into the fluid, which is discarded. Glucose, electrolytes, and drugs can be administered by adding them to the dialysis fluid so they will diffuse through the membrane into the blood. People with renal insufficiency accumulate substantial amounts of excess body water between treatments, and dialysis serves also to remove it. Hemodialysis patients typically have three sessions per week for 4 to 8 hours per session. In addition to inconvenience, hemodialysis carries risks of infection and thrombosis. Blood tends to clot when exposed to foreign surfaces, so an anticoagulant such as heparin is added during dialysis. It can be carried out at home by the patient, who is provided with plastic bags of dialysis fluid. The fluid is left in the body cavity for 15 to 60 minutes to allow the blood to equilibrate with it; then it is drained, discarded, and replaced with fresh dialysis fluid. The patient is not limited by a stationary dialysis machine and can go about most normal activities. Blood is Blood pump pumped into a dialysis chamber, where it flows through a selectively permeable membrane surrounded by dialysis fluid. Structure of the glomerular filtration membrane; roles of the capillary fenestrations, basement membrane, and filtration slits in producing glomerular filtrate; the difference between the filtrate and blood plasma 3. Structure and function of the juxtaglomerular apparatus and how tubuloglomerular feedback works 9. Effects of antidiuretic hormone and aldosterone on the kidney and how they reduce water loss from the body 5. Effects of aldosterone, natriuretic peptides, parathyroid hormone, calcitonin, and calcitriol on nephron function Assess Your Learning Outcomes To test your knowledge, discuss the following topics with a study partner or in writing, ideally from memory. Four main nitrogenous wastes and their metabolic sources; how metabolic wastes differ from other wastes 4. Blood urea nitrogen, azotemia, and uremia, and the pathological effects of uremia 5. Arrangement of the renal parenchyma around the renal sinus; organization of the cortex, medulla, and renal pyramids 5. The relationship of a renal pyramid to a minor calyx, and of the minor calyces to the major calyces, renal pelvis, and ureter 6. The term for the microscopic functional units of the kidney, and their approximate number per kidney 7. Blood flow from the renal artery through the kidney to the renal vein, including circulation through both the cortex and the medulla 8. Fluid flow from the point where it filters from the blood to the point where it leaves the kidney; those parts of the renal tubule that belong to a single nephron and the part that is shared by multiple nephrons 10. The distinction between glomerular filtrate, tubular fluid, and urine in relation to the progress of the fluid through the renal tubule 11. Differences in the structure and function of cortical nephrons and juxtamedullary nephrons 12. Innervation of the kidney and two effects of sympathetic stimulation on renal function 23. Why urine is yellow; why the shade of yellow varies; and some causes of other, unusual colors 2. The three most abundant solutes in urine, and some causes of other, unusual solutes 5. The defining sign of diabetes in general; four forms of diabetes and their causes 7. The general effect of diuretics; modes of diuretic action of caffeine, alcohol, and loop diuretics 8. Transcellular and paracellular routes of reabsorption, and the role of solvent drag 3. How tubular reabsorption is limited by the transport maximum (Tm); how this relates to glycosuria in diabetes mellitus 23. Anatomy of the male urethra, its three segments, and the internal and external urethral sphincters 5. Anatomy and histology of the urinary bladder; the detrusor, mucosal epithelium, rugae, and trigone 3. Anatomy of the female urethra and external urethral sphincter Testing Your Recall 1. The two ureters and the urethra form the boundaries of a smooth area called the on the floor of the urinary bladder. The is a group of epithelial cells of the nephron loop that monitors the composition of the tubular fluid. To enter the capsular space, filtrate must pass between foot processes of the, cells that form the visceral layer of the glomerular capsule. Glycosuria occurs if the rate of glomerular filtration of glucose exceeds the of the proximal convoluted tubule. Beavers have relatively little need to conserve water and could therefore be expected to have than humans do. The sphincter of the male is under involuntary control and relaxes during the micturition reflex. Very little is found in the glomerular filtrate because it is negatively charged and is repelled by the basement membrane of the glomerulus.
The vasa recta are arranged as a countercurrent exchange system that enables them to remove water from the medulla without subtracting from its osmotic gradient androgen hormone quizzes cheap 60 caps pilex with amex. Concisely contrast the role of the countercurrent multiplier with that of the countercurrent exchanger prostate cancer psa levels proven pilex 60 caps. Urinalysis prostate cancer for dummies purchase pilex amex, the examination of the physical and chemical properties of urine androgen hormone sensitivity purchase pilex with paypal, is therefore one of the most routine procedures in medical examinations androgen hormone of happiness discount pilex 60 caps buy online. The principal characteristics of urine and certain tests used to evaluate renal function are described here. The yellow color of urine is due to urochrome,18 a pigment produced by the breakdown of hemoglobin from expired erythrocytes. Pink, green, brown, black, and other colors can result from certain foods, vitamins, drugs, and metabolic diseases. Urine is normally clear but turns cloudy upon standing because of bacterial growth. Blood in the urine (hematuria) may be due to a urinary tract infection, trauma, or kidney stones. Cloudiness or blood in a urine specimen sometimes, however, simply indicates contamination with semen or menstrual fluid. As it stands, however, bacteria multiply, degrade urea to ammonia, and produce the pungent odor typical of stale wet diapers. This is a ratio of the density (g/mL) of a substance to the density of distilled water. Multiplying the last two digits of the specific gravity by a proportionality constant of 2. Urine can have an osmolarity as low as 50 mOsm/L in a very hydrated person or as high as 1,200 mOsm/L in a dehydrated person. Compared with the osmolarity of blood (300 mOsm/L), then, urine can be either hypotonic or hypertonic. The body constantly generates metabolic acids and gets rid of them by excreting mildly acidic urine, usually with a pH of about 6. Normally, the most abundant solute is urea, followed by sodium chloride, potassium chloride, and lesser amounts of creatinine, uric acid, phosphates, sulfates, and traces of calcium, magnesium, and sometimes bicarbonate. It is abnormal to find glucose, free hemoglobin, albumin, ketones, or bile pigments in the urine; their presence is an important indicator of disease. Fluid intake and some drugs can temporarily increase output to as much as 20 L/day. Low output can result from kidney disease, dehydration, circulatory shock, prostate enlargement, and other causes. If urine output drops to <400 mL/day, the body cannot maintain a safe, low concentration of wastes in the blood plasma. Here we examine two methods used to determine renal clearance and glomerular filtration rate. There are at least four forms of diabetes: diabetes mellitus24 type 1 and type 2, gestational diabetes, and diabetes insipidus. Glucose osmotically retains water in the tubule, so more water passes in the urine (osmotic diuresis) and a person may become severely dehydrated. In diabetes mellitus and gestational diabetes, the high glucose level in the tubular fluid is a result of hyperglycemia, a high level in the blood. Tests for glycosuria are now as simple as dipping a chemical test strip into the urine specimen-an advance in medical technology for which urologists are no doubt grateful. In diabetes insipidus, the urine contains no glucose and, by the old diagnostic method, would not taste sweet. Renal Clearance Renal clearance is the volume of blood plasma from which a particular waste is completely removed in 1 minute. It represents the net effect of three processes: Glomerular filtration of the waste + Amount added by tubular secretion Amount removed by tubular reabsorption Renal clearance In principle, we could determine renal clearance by sampling blood entering and leaving the kidney and comparing their waste concentrations. This means the equivalent of 60 mL of blood plasma is completely cleared of urea per minute. This is a normal rate of urea clearance and is sufficient to maintain safe levels of urea in the blood. Some diuretics, such as caffeine, act by dilating the afferent arteriole and thus increasing glomerular filtration rate. Loop diuretics such as furosemide (Lasix) act on the nephron loop to inhibit the Na+K+Cl symport. This impairs the countercurrent multiplier, thus reducing the osmotic gradient in the renal medulla and making the collecting duct unable to reabsorb as much water as usual. All inulin filtered by the glomerulus remains in the renal tubule and appears in the urine; none is reabsorbed, nor does the tubule secrete it. This has a small but acceptable error of measurement, and is an easier procedure than injecting and measuring inulin. This is made possible by an apparatus for storing urine and neural controls for its timely release. The ureters pass posterior to the bladder and enter it from below, passing obliquely through its muscular wall and opening onto its floor. A small flap of mucosa acts as a valve at the opening of each ureter into the bladder, preventing urine from backing up into the ureter when the bladder contracts. The adventitia is a connective tissue layer that binds it to the surrounding tissues. The muscularis consists of two layers of smooth muscle over most of its length, but a third layer appears in the lower ureter. The mucosa has a urothelium (transitional epithelium) that begins in the minor calyces of the kidney and extends from there through the bladder. When urine enters the ureter and stretches it, the muscularis contracts and initiates a peristaltic wave that milks the urine from the renal pelvis down to the bladder. These contractions occur every few seconds to few minutes, proportional to the rate at which urine enters the ureter. The lumen of the ureter is very narrow and is easily obstructed or injured by kidney stones (see Deeper Insight 23. It is covered by parietal peritoneum on its flattened superior surface and by a fibrous adventitia elsewhere. Renal calculi form in the renal pelvis and are usually small enough to pass unnoticed in the urine flow. Some, however, grow as large as a few centimeters and block the renal pelvis or ureter, which can lead to the destruction of nephrons as pressure builds in the kidney. A large, jagged calculus passing down the ureter stimulates strong contractions that can be excruciatingly painful. Causes of renal calculi include hypercalcemia (excess calcium in the blood), dehydration, pH imbalances, frequent urinary tract infections, or an enlarged prostate gland causing urine retention. The patient lies on an operating table under general anesthesia with a water-filled cushion on the abdomen or behind the kidney. One to two thousand underwater electrical sparks produce focused shock waves that pulverize the calculi into "stone dust" that the patient can subsequently pass in the urine. The mucosa is lined with urothelium (transitional epithelium), whose unique surface umbrella cells protect it from the hypertonic and acidic urine as described in section 5. The epithelium is thicker here than anywhere else in the urinary tract, since it is subject to the most prolonged exposure to stored urine. As the bladder fills, the rugae flatten and the epithelium thins to two or three cells thick. Blood flows through the arteries just before entering the cortical radiate arteries. Tight junctions prevent material from leaking between the epithelial cells of the renal tubule. If all other conditions remain the same, dilation of the efferent arteriole raises the glomerular filtration rate. The minimum osmolarity of urine is 300 mOsm/L, equal to the osmolarity of the blood. The amount of glucose in the urine is directly proportional to the amount of sodium. How would the glomerular filtration rate be affected by kwashiorkor (see Deeper Insight 18. Discuss how the unity of form and function is exemplified by each of the following comparisons: (a) the thin and thick segments of the nephron loop; (b) the proximal and distal convoluted tubules; and (c) the afferent and efferent arterioles. The flow of matter down osmotic, concentration, and electrochemical gradients is important in several processes in this chapter (see section 1. You must be familiar with the role of electrolytes in the membrane potentials of cells in order to understand the effects of electrolyte imbalances on nerve and muscle function (see section 12. Electrolyte concentrations are expressed in milliequivalents per liter (mEq/L), a unit of measurement explained in appendix B. Effects of the respiratory rhythm on blood Pco2 and pH and, conversely, the effects of these variables on respiration, are central to understanding acidbase balance (see section 22. A good knowledge of nephron function is vital for understanding the role of the kidneys in fluid, electrolyte, and acidbase balance (see sections 23. We enter the world in rather soggy condition, having swallowed, excreted, and floated in amniotic fluid for months. Young adult men average 55% to 60% water; women average slightly less because they have more adipose tissue, which is nearly free of water. Fluid is continually exchanged between compartments by way of capillary walls and plasma membranes (fig. Water moves by osmosis from the digestive tract to the bloodstream and by capillary filtration from the blood to the tissue fluid. From the tissue fluid, it may be reabsorbed by the capillaries, osmotically absorbed into cells, or taken up by the lymphatic system, which returns it to the bloodstream. If a local imbalance arises, osmosis usually restores the balance within seconds so that intracellular and extracellular osmolarity are equal. If the osmolarity of the tissue fluid rises, water moves out of the cells; if it falls, water moves into the cells. Osmosis from one fluid compartment to another is determined by the relative concentration of solutes in each compartment. C ellular function requires a fluid medium with a carefully controlled composition. If the quantity, osmolarity, electrolyte concentration, or pH of this medium is altered, life-threatening disorders of cellular function may result. Consequently, the body has several mechanisms for keeping these variables within narrow limits and maintaining three types of homeostatic stability: fluid, electrolyte, and acidbase balance. These balances are maintained by the collective action of the urinary, respiratory, digestive, integumentary, endocrine, nervous, cardiovascular, and lymphatic systems. This chapter describes the homeostatic regulation of fluid, electrolyte, and acidbase balance and shows the close relationship of these variables to each other. These balances are so crucial that fluid therapy, aimed at restoring one or more of these, is often a critical aspect of patient care. Problems of fluid and electrolyte balance also arise frequently in sports and recreational medicine, in situations ranging from summer football practice to backcountry hiking. There is a two-way exchange of water between the blood and tissue fluid and between the tissue fluid and intracellular fluids. Excess tissue fluid is picked up by the lymphatic system, which returns it to the bloodstream. Gains 2,500 mL/day Losses 2,500 mL/day the other source is preformed water, which is ingested in food (700 mL/day) and drink (1,600 mL/day). You can easily · · Metabolic water 200 mL Feces 200 mL Expired air 300 mL visualize this by breathing onto a cool surface such as a mirror. A simple way to observe it is to cup the palm of your hand for a minute against a cool nonporous surface such as a laboratory benchtop or mirror. When you take your hand away, you will notice the water that transpired through the skin and condensed on that surface, even in places that were not in contact with your skin. Food 700 mL Cutaneous transpiration 400 mL Sweat 100 mL Drink 1,600 mL Urine 1,500 mL Water loss varies greatly with physical activity and environmental conditions. Respiratory loss increases in cold weather, for example, because cold air is drier and absorbs more body water from the respiratory tract. Prolonged, heavy work can raise the respiratory loss to 650 mL/day and perspiration to as much as 5 L/day, though it reduces urine output by nearly two-thirds. Sensible water loss is noticeable output, particularly through the urine and in case of sufficient sweating to produce obvious wetness of the skin. Obligatory water loss is output that is relatively unavoidable: expired air, cutaneous transpiration, sweat, fecal moisture, and the minimum urine output, about 400 mL/day, needed to prevent azotemia. Even dehydrated individuals cannot prevent such losses; thus, they become further dehydrated. Thirst Sense of thirst Ingestion of water Cools and moistens mouth Distends Short-term stomach inhibition and intestines of thirst Long-term inhibition of thirst Stimulates hypothalamic osmoreceptors 24. A mere 2% to 3% increase in plasma osmolarity makes a person intensely thirsty, as does a 10% to 15% blood loss. There are two reasons for this: (1) the osmoreceptor response leads to sympathetic output from the hypothalamus that inhibits the salivary glands. Reduced salivation produces a dry, sticky-feeling mouth and a desire to drink, but it is by no means certain that this is the primary motivation to drink. Long-term satiation of thirst depends on absorbing water from the small intestine and lowering the osmolarity of the blood. Reduced osmolarity stops the osmoreceptor response, promotes capillary filtration, and makes the saliva more abundant and watery. However, these changes require 30 minutes or longer to take effect, and it would be rather impractical if we had to drink that long while waiting to feel satisfied. Fortunately, there are mechanisms that act more quickly to temporarily quench the thirst and allow time for the change in blood osmolarity to occur. One of these is cooling and moistening the mouth; rats drink less if their water is cool than if it is warm, and simply moistening the mouth temporarily satisfies an animal even if the water is drained from its esophagus before it reaches the stomach.
Other traits commonly regarded as male secondary sex characteristics are the facial hair androgen hormone x organic discount pilex 60 caps buy, relatively coarse and visible hair on the torso and limbs prostate cancer testosterone buy cheap pilex 60 caps on line, and the relatively muscular physique prostate cancer 911 commission report pilex 60 caps order on-line. In females prostate 22 cheap pilex 60 caps without a prescription, they include the distribution of body fat prostate cancer 999 proven 60 caps pilex, enlargement of the breasts (independently of lactation), and relatively hairless appearance of the skin. The distinction begins with the combination of sex chromosomes bequeathed to the zygote. Most of our cells have 23 pairs of chromosomes: 22 pairs of autosomes and 1 pair of sex chromosomes (see fig. Every egg contains an X chromosome, but half of the sperm carry an X and the other half carry a Y. Even an adult male, however, retains a tiny Y-shaped vestige of the paramesonephric ducts, like a vestigial uterus and uterine tubes, in the area of the prostatic urethra. It may seem as if androgens should induce the formation of a male reproductive tract and estrogens induce a female reproductive tract. However, the estrogen level is always high during pregnancy, so if this mechanism were the case, it would feminize all fetuses. Thus, the development of a female results from the absence of androgens, not the presence of estrogens. The sex of a child is determined by whether the egg is fertilized by an X-bearing sperm or a Y-bearing sperm. In the embryo, the genitals begin developing from identical structures in both sexes. It requires an interaction between genes and hormones produced by the mother and fetus. Just as we have seen with other hormones, those involved here require specific receptors on their target cells to exert an effect. Up to a point, a fetus is sexually undifferentiated, or "noncommittal" as to which sex it will become. Its gonads begin to develop at 5 to 6 weeks as gonadal ridges, each lying alongside a primitive kidney, the mesonephros, which later degenerates. In males, the mesonephric ducts develop into the reproductive tract and the paramesonephric ducts degenerate. By 8 to 9 weeks, the male gonadal ridge has become a rudimentary testis that begins to secrete testosterone. Testosterone stimulates the mesonephric duct on its own side to develop into the system of male reproductive posterior to the genital tubercle; and labioscrotal folds, a larger pair of tissue folds lateral to the urogenital folds. By the end of week 9, the fetus begins to show sexual differentiation, and either male or female genitalia are distinctly formed by the end of week 12 (fig. In the female, the three structures just listed become the clitoral glans, labia minora, and labia majora, respectively; all of these are more fully described in the next chapter. In the male, the genital tubercle elongates to form the phallus; the urogenital folds fuse to enclose the urethra, joining the phallus to form the penis; and the labioscrotal folds fuse to form the scrotum, a sac that will later contain the testes. Male and female organs that develop from the same embryonic structure are said to be homologous. Thus, the penis is homologous to the clitoris and the scrotum is homologous to the labia majora. In the presence of excess androgen, the clitoris may become greatly enlarged and resemble a small penis. In other cases, the ovaries descend into the labia majora as if they were testes descending into a scrotum. Such abnormalities sometimes result in mistaken identification of the sex of an infant at birth. Note that the male tract develops from the mesonephric duct and the female tract from the paramesonephric duct; the other duct in each sex degenerates. As the male gubernaculum continues to grow, it passes between the internal and external oblique muscles and into the scrotal swelling. Independently of migration of the testis, the peritoneum also develops a fold that extends into the scrotum as the vaginal 7 process. By 6 weeks, the embryo has three primordial structures-the genital tubercle, urogenital folds, and labioscrotal folds-which will become the male or female genitalia. At 8 weeks, these structures have grown but the sexes are still indistinguishable. Slight sexual differentiation is noticeable at 10 weeks, and the sexes are fully distinguishable by 12 weeks. An extension of the peritoneum called the vaginal process passes through the inguinal canal and becomes the tunica vaginalis. The superior part of the embryonic gonad degenerates and its inferior part migrates downward, guided by the gubernaculum. In the seventh month, the testes abruptly pass through the inguinal canals into the scrotum. As they descend, they are accompanied by ever-elongating testicular arteries and veins and by lymphatic vessels, nerves, spermatic ducts, and extensions of the internal oblique muscle. The vaginal process becomes separated from the peritoneal cavity and persists as a sac, the tunica vaginalis, enfolding the anterior and lateral sides of the testis and protecting the testis from abrasion against the scrotum with movements of the body. The actual mechanism of descent remains obscure, although multiple hypotheses have been offered. The reason this descent is necessary, however, will be explained in the next section. Uncorrected cryptorchidism, however, leads inevitably to sterility and sometimes to testicular cancer. The female gubernaculum extends from the inferior pole of the ovary to the labioscrotal fold. We will survey the male reproductive system in order according to the sites of sperm formation, transport, and emission-therefore beginning with the scrotum and testes, continuing through the spermatic ducts and accessory glands associated with them, and ending with the penis. The scrotum10 is a pendulous pouch of skin, muscle, and fibrous connective tissue, containing the testes (fig. The skin has sebaceous glands, sparse hair, rich sensory innervation, and somewhat darker pigmentation than skin elsewhere. Just deep to the skin is a layer of smooth muscle and connective tissue called the dartos11 fascia; its muscle is called the dartos muscle. The scrotum is divided into right and left compartments by an internal median septum, which prevents infections of one testis from spreading to the other one. Posteriorly, the scrotum contains the spermatic cord, a bundle of fibrous connective tissue containing the ductus deferens (a sperm duct), lymphatic vessels, testicular nerves, the testicular artery, and a complex of veins called the pampiniform12 plexus closely surrounding the artery. It continues across the anterior side of the pubis and into a 4 cm inguinal canal, which leads through the muscles of the groin and emerges into the pelvic cavity. The inferior entrance into the inguinal canal is called the superficial inguinal ring, and its superior exit into the pelvic cavity is the deep inguinal ring. Within the scrotum, the spermatic cord is enmeshed in strips of internal oblique muscle called the cremaster. When it is cold, the cremaster contracts and draws the testes closer to the body to keep them warm. When it is warm, the cremaster relaxes and the testes are suspended farther from the body. This, too, contracts when it is cold, and the scrotum becomes taut and even more wrinkled. The tautness of the scrotum helps to hold the testes snugly against the warm body and it reduces the surface area of the scrotum, thus reducing heat loss. Without this venous plexus, warm arterial blood would heat the testis and inhibit sperm production. The pampiniform plexus, however, prevents this by acting as a countercurrent heat exchanger (fig. The relatively cool venous blood ascending the spermatic cord toward the pelvic cavity draws heat out of the descending arterial blood headed for the testis, so by the time it reaches the testis the blood is 1. Although illustrated as one vessel, the pampiniform plexus is a network of veins surrounding the testicular artery (see fig. Note the cooling of the arterial blood as it descends the artery and its heat transfers to the adjacent, ascending venous blood. The testis is oval and slightly flattened, about 4 cm long, 3 cm from anterior to posterior, and 2. Connective tissue septa extend from the capsule into the parenchyma, dividing it into 250 to 300 wedge-shaped lobules. Between the seminiferous tubules are clusters of interstitial16 endocrine cells, the source of testosterone. The epithelium consists of several layers of germ cells in the process of becoming sperm, and a much smaller number of tall nurse cells (supporting cells, sustentocytes17), which protect the germ cells and promote their development. The germ cells depend on the nurse cells for nutrients, waste removal, growth factors, and other needs. The nurse cells also secrete two proteins called androgen-binding protein and inhibin, both of which regulate sperm production in ways described later. A nurse cell is shaped a little like a tree trunk whose roots spread out over the basement membrane, forming the boundary of the tubule, and whose thick trunk reaches to the tubule lumen. This is important because the germ cells, being genetically different from other cells of the body, would otherwise be attacked by the immune system. They are moved along by the flow of fluid secreted by the nurse cells and by the cilia on some rete cells. Each testis is supplied by a testicular artery that arises from the abdominal aorta just below the renal artery. This is a very long, slender artery that winds its way down the posterior abdominal wall before passing through the inguinal canal into the scrotum (see fig. Its blood pressure is very low, and indeed this is one of the few arteries to have no pulse. Consequently, blood flow to the testes is quite meager and the testes receive a poor oxygen supply. In response to this, the sperm develop unusually large mitochondria, which may precondition them for survival in the hypoxic environment of the female reproductive tract. As these veins pass through the inguinal canal, they converge and form the testicular vein. The right testicular vein drains into the inferior vena cava and the left one drains into the left renal vein. They travel through the inguinal canal with the veins and lead to lymph nodes adjacent to the lower aorta. Lymph from the penis and scrotum, however, travels to lymph nodes adjacent to the iliac arteries and veins and in the inguinal region. They are mixed sensory and motor nerves containing predominantly sympathetic but also some parasympathetic fibers. The sensory fibers are concerned primarily with pain and the autonomic fibers are predominantly vasomotor, for regulation of blood flow. The duct is about 6 m (20 ft) long, but it is so slender and highly coiled that it is packed into an epididymis only 7. Sperm are physiologically immature when they leave the testis but mature as they travel through the head and body of the epididymis. They are stored here and in the adjacent portion of the ductus deferens pending ejaculation, and remain viable for 40 to 60 days. If they become too old without being ejaculated, they disintegrate and the epididymis reabsorbs them. It passes upward through the spermatic cord and inguinal canal and enters the pelvic cavity. After passing between the bladder and ureter, the duct turns downward behind the bladder and widens into a terminal ampulla. The ductus deferens ends by uniting with the duct of an accessory gland, the seminal vesicle. The duct has a very narrow lumen and a thick wall of smooth muscle well innervated by sympathetic nerve fibers. Vasectomy, the surgical method of male contraception, consists of cutting out a short portion of the ductus (vas) deferens to interrupt the passage of sperm. Where the ductus deferens and duct of the seminal vesicle meet, they form a short (2 cm) ejaculatory duct, which passes through the prostate and empties into the urethra. These include the following: the male urethra is shared by the reproductive and urinary systems. It is about 18 cm long and consists of three regions: the prostatic, membranous, and spongy (penile) urethra. Although it serves both urinary and reproductive roles, it cannot pass urine and semen simultaneously for reasons explained in section 27. The seminal vesicles (seminal glands) are a pair of glands posterior to the urinary bladder; one is associated with each ductus deferens. The secretory portion is a very convoluted duct with numerous branches that form a complex labyrinth. The yellowish secretion of the seminal vesicles constitutes about 65% to 75% of the semen. About 12 small efferent ductules arise · from the posterior side of the testis and carry sperm to the epididymis. The superior clublike portion where it receives the efferent ductules is called the head; the long middle portion below the last efferent ductule is the body; and the slender inferior end is the tail. It measures about 2 × 4 × 3 cm and is an aggregate of 30 to 50 compound tubuloacinar glands enclosed in a single fibrous capsule. The stroma of the prostate consists of connective tissue and smooth muscle, like that of the seminal vesicles.
The venous palmar arches mens health store order cheap pilex on-line, and brachial mens health magazine australia purchase pilex 60 caps with visa, basilic man health over 50 discount pilex 60 caps buy on-line, axillary mens health 3 bean chili purchase 60 caps pilex with mastercard, and subclavian veins (section 20 uw prostate oncology center discount pilex online mastercard. Continuation of the external iliac artery as the femoral artery; deep femoral and circumflex femoral branches of the femoral artery; popliteal artery; and anterior and posterior tibial arteries (section 20. The dorsal pedal and arcuate arteries that arise from the anterior tibial artery; fibular, medial plantar, and lateral plantar arteries; and deep plantar arch (section 20. The superficial dorsal venous arch, small and great saphenous veins, and popliteal vein (section 20. The deep plantar venous arch; the lateral and medial plantar veins; fibular and posterior tibial veins; and anterior tibial veins (section 20. For all named blood vessels in this outline, their anatomical location; the vessel from which they arise; the course they follow; and the organs, body regions, or other blood vessels they supply 2. The ascending aorta, aortic arch, and descending aorta, and the thoracic and abdominal segments of the descending aorta (section 20. Branches that arise from the ascending aorta and aortic arch: the coronary arteries, brachiocephalic trunk, left common carotid artery, and left subclavian artery (section 20. Four principal arteries of the neck: the common carotid, vertebral artery, thyrocervical trunk, and costocervical trunk (section 20. The external and internal carotid arteries; branches of the external carotid (superior thyroid, lingual, facial, occipital, maxillary, and superficial temporal arteries); and branches of the internal carotid (ophthalmic, anterior cerebral, and middle cerebral arteries) (section 20. Convergence of the vertebral arteries to form the basilar artery; the posterior cerebral arteries and arteries to the cerebellum, pons, and inner ear arising from the basilar artery (section 20. In a case where interstitial hydrostatic pressure is negative, the only force causing capillaries to reabsorb fluid is a. The brain receives blood from all of the following vessels except the artery or vein. The highest arterial blood pressure attained during ventricular contraction is called pressure. A blood vessel adapted to withstand a high pulse pressure would be expected to have a. A person with a systolic blood pressure of 130 mm Hg and a diastolic pressure of 85 mm Hg would have a mean arterial pressure of about a. The difference between the colloid osmotic pressure of blood and that of the tissue fluid is called. Movement across the capillary endothelium by the uptake and release of fluid droplets is called. All efferent fibers of the vasomotor center belong to the division of the autonomic nervous system. Most of the blood supply to the brain comes from a ring of arterial anastomoses called the. The major superficial veins of the arm are the on the medial side and on the lateral side. The lungs receive blood exclusively from the pulmonary circuit of the circulatory system. In the baroreflex, a drop in arterial blood pressure triggers a corrective vasodilation of the systemic blood vessels. If the radius of a blood vessel doubles and all other factors remain the same, blood flow through that vessel also doubles. The femoral triangle is bordered by the inguinal ligament, sartorius muscle, and rectus femoris muscle. Blood cannot get from an artery to a vein without first passing through some capillaries. Aldosterone secreted by the adrenal gland must be delivered to the kidney immediately below. Trace the route that an aldosterone molecule must take from the adrenal gland to the kidney, naming all major blood vessels in the order traveled. People in shock commonly exhibit paleness, cool skin, tachycardia, and a weak pulse. Discuss why it is advantageous to have baroreceptors in the aortic arch and carotid sinus rather than in some other location such as the common iliac arteries. Brushing up on the meaning of those terms and their structural relationships may help you better understand anatomy of the lymphatic organs (see section 5. The mechanisms of lymph flow are similar to those for the venous return of blood (see section 20. You can brush up on leukocyte types, appearances, and functions most easily in table 18. The actions of immune cells against disease agents involve the processes of phagocytosis, receptor-mediated endocytosis, and exocytosis described in section 3. Indeed, he shared the 1908 Nobel Prize for Physiology or Medicine with Paul Ehrlich (18541915), who had developed the theory of humoral immunity, a process also discussed in this chapter. This chapter focuses largely on the immune system, which is not an organ system but rather a population of cells that inhabit all of our organs and defend the body from agents of disease. But immune cells are especially concentrated in a true organ system, the lymphatic system. This is a network of organs and veinlike vessels that recover tissue fluid, inspect it for disease agents, activate immune responses, and return the fluid to the bloodstream. After all, human homeostasis works wonderfully not only to sustain our lives, but also to provide a predictable, warm, wet, nutritious habitat for our internal guests. Many of these guest microorganisms are beneficial or even necessary to human health, but some have the potential to cause disease if they get out of hand. One of these defenses was discovered in 1882 by a moody, intense, Russian zoologist, Elie Metchnikoff (18451916). When studying the tiny transparent larvae of starfish, he observed mobile cells wandering throughout their bodies. He thought at first that they must be digestive cells, but when he saw similar cells in sea anemones ingest nonnutritive dye particles that he injected, he thought they must play a defensive role. Metchnikoff knew that mobile cells also exist in human blood and pus and quickly surround a splinter introduced through the skin, so he decided to experiment to see if the starfish cells would do the same. He impaled a starfish larva on a rose thorn, and the next morning he found the thorn crawling with cells that seemed to be trying to devour it. He later saw similar cells devouring and digesting infectious yeast in tiny transparent crustaceans called water fleas. He coined the word phagocytosis for this reaction and named the wandering cells phagocytes-terms we still use today. Metchnikoff showed that animals from simple sea anemones and starfish to humans actively defend themselves against disease agents. His observations marked the founding of cellular and comparative immunology, and won him the scientific respect the lymphatic system (fig. Fluid continually filters from blood capillaries into the tissue (interstitial) spaces. One would die of circulatory failure within hours if this water and protein were not returned to the bloodstream. One task of the lymphatic system is to reabsorb this excess and return it to the blood. Even partial interference with lymphatic drainage can lead to severe lymphedema (see fig. As the lymphatic system recovers tissue fluid, it also picks up foreign cells and chemicals from the tissues. On its way back to the bloodstream, the fluid passes through lymph nodes, where immune cells stand guard against foreign matter. When they detect anything potentially harmful, they activate a protective immune response. In the small intestine, special lymphatic vessels called lacteals absorb dietary lipids that are not absorbed by the blood capillaries. After a meal, for example, lymph draining from the small intestine has a milky appearance because of its lipid content. Lymph leaving the lymph nodes contains a large number of lymphocytes-indeed, this is the main supply of lymphocytes to the bloodstream. Lymph may also contain macrophages, hormones, bacteria, viruses, cellular debris, or even traveling cancer cells. These begin with microscopic lymphatic capillaries (terminal lymphatics), which penetrate nearly every tissue of the body but are absent from cartilage, bone, bone marrow, and the cornea. They are closely associated with blood capillaries, but unlike them, they are closed at one end (fig. A lymphatic capillary consists of a sac of thin endothelial cells that loosely overlap each other like the shingles of a roof. The cells are tethered to surrounding tissue by anchoring filaments that prevent the sac from collapsing. Thus, the composition of lymph arriving at a lymph node is like a report on the state of the upstream tissues. The overlapping edges of the endothelial cells act as valves that can open and close. When tissue fluid pressure is high, it pushes the flaps inward (open) and fluid flows into the capillary. When pressure is higher in the lymphatic capillary than in the tissue fluid, the flaps are pressed outward (closed). Explain why their structural difference is related to their functional difference. Their walls are thinner and their valves are closer together than those of the veins. As the lymphatic vessels converge along their path, they become larger and larger vessels with changing names. The route from the tissue fluid back to the bloodstream is: lymphatic capillaries collecting vessels 11 lymphatic trunks two collecting ducts subclavian veins. Thus, there is a continual recycling of fluid from blood to tissue fluid to lymph and back to the blood (fig. These often travel alongside veins and arteries and share a common connective tissue sheath with them. The lymph trickles slowly through each node, where bacteria are phagocytized and immune cells monitor the fluid for foreign antigens. The lymphatic system picks up excess tissue fluid and returns it to the bloodstream. Eventually, the collecting vessels converge to form larger lymphatic trunks, each of which drains a major portion of the body. There are 11 of these, whose names indicate their locations and parts of the body they drain: a solitary intestinal trunk and paired jugular, subclavian, bronchomediastinal, intercostal, and lumbar trunks. The lymphatic trunks converge to form two collecting ducts, the largest of the lymphatic vessels (fig. The right lymphatic duct is formed by the convergence of the right jugular, subclavian, and bronchomediastinal trunks in the right thoracic cavity. It receives lymphatic drainage from the right arm and right side of the thorax and head and empties into the right subclavian vein. Identify two benefits in having lymphatic capillaries pick up tissue fluid that is not reclaimed by the blood capillaries. It begins just below the diaphragm anterior to the vertebral column at the level of the second lumbar vertebra. The thoracic duct then passes through the diaphragm with the aorta and ascends the mediastinum adjacent to the vertebral column. As it passes through the thorax, it receives additional lymph from the left bronchomediastinal, left subclavian, and left jugular trunks, then empties into the left subclavian vein. Collectively, this duct therefore drains all of the body below the diaphragm, and the left upper limb and left side of the head, neck, and thorax. Why are the axillary lymph nodes often biopsied in cases of suspected breast cancer Flow of Lymph Lymph flows under forces similar to those that govern venous return (see section 20. The primary mechanism of flow is rhythmic contractions of the lymphatic vessels themselves, which contract when the fluid stretches them. The valves of lymphatic vessels, like those of veins, prevent the fluid from flowing backward. Lymph flow is also produced by skeletal muscles squeezing the lymphatic vessels, like the skeletal muscle pump that moves venous blood. Since lymphatic vessels are often wrapped with an artery in a common connective tissue sheath, arterial pulsation also rhythmically squeezes the adjacent lymphatic vessels and contributes to lymph flow. A thoracic (respiratory) pump promotes the flow of lymph from the abdominal to the thoracic cavity as one inhales, just as it does in venous return. Finally, at the point where the collecting ducts empty into the subclavian veins, the rapidly flowing bloodstream draws the lymph into it. These tissues are composed of a variety of lymphocytes and other cells with various roles in defense and immunity: 1. Neutrophils are aggressively antibacterial leukocytes and are described in section 18. T lymphocytes (T cells) are lymphocytes that mature in the thymus and later depend on thymic hormones; the T stands for thymus-dependent. B lymphocytes (B cells) are lymphocytes that differentiate into plasma cells-connective tissue cells that secrete antibodies. They are named for an organ in chickens (the bursa of Fabricius1) in which they were first discovered.
Buy pilex with paypal. Fat-Blasting Battle Rope 10-Min Workout | BURNER | Men's Health.
References
- Goldfischer ER, Clayton AH, Goldstein I, et al: Decreased sexual desire screener (DSDS) for diagnosis of hypoactive sexual desire disorder in women, Obstet Gynecol 111:109, 2008.
- Finkelstein JB, Levy AC, Silva MV, et al: How to decide which infant can have robotic surgery? Just do the math, J Pediatr Urol 11(4):170.e1-170. e4, 2015.
- Reis-Filho JS, Ellis I. Invasive micropapillary carcinoma. In: Lakhani S, Ellis IO, Schnitt SJ, Tan P-H, van de Vijver MJ, eds. WHO Classification of Tumors of the Breast. Lyon: IARC Press 2012;65-66.
- Berk DR, Varich LJ. Localized persistent pulmonary interstitial emphysema in a preterm infant in the absence of mechanical ventilation. Pediatr Radiol 2005;35:1243-5.
- Sigurdsson L, Reyes J, Kocoshis SA, et al. Bacteremia after intestinal transplantation in children correlates temporally with rejection or gastrointestinal lymphoproliferative disease. Transplantation. 2002;70:302-305.
- Guidelines for the management of adults with hospital-acquired, ventilatorassociated, and healthcare-associated pneumonia. Am J Respir Crit Care Med 171:388, 2005.
