
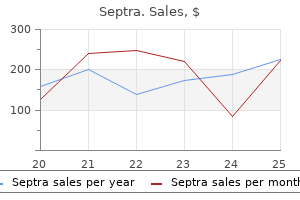
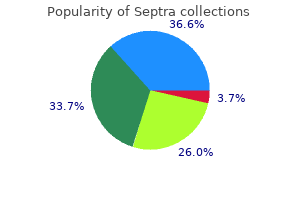
Septra
| Contato
Página Inicial
Nicole DePasquale, PhD
- Assistant Professor in Medicine
https://medicine.duke.edu/faculty/nicole-depasquale-phd
The chromosomes of each homologous pair are alike in size and shape and contain genes for the same traits symptoms quotes buy genuine septra on-line. Sperm cells and oocytes contain the haploid (haployd) number (n) of chromosomes treatment narcolepsy purchase 480mg septra otc, which is half the diploid number medications 4h2 discount septra 480mg online, or 23 medicine for constipation septra 480mg buy low price. Reduction in the number of chromosomes in sperm cells or oocytes to an n number is important symptoms 0f high blood pressure purchase septra 480 mg with mastercard. When a sperm cell and an oocyte fuse to form a fertilized egg, each provides an n number of chromosomes, which reestablishes a 2n number. If meiosis did not take place, the number of chromosomes in the fertilized oocyte would double each time fertilization occurred, and the extra chromosomal material would be lethal to the developing offspring. The baby is male if the oocyte is fertilized by a Y-carrying sperm cell or female if it is fertilized by an X-carrying sperm cell. The stages of meiosis have the same names as the stages of mitosis-that is, prophase, metaphase, anaphase, and telophase-but distinct differences exist between mitosis and meiosis. At the beginning of meiosis, each of the 46 chromosomes consists of two sister chromatids (krm-tids) connected by a centromere (sentr-mr; figure 28. In prophase I, the chromosomes become visible, and the homologous pairs come together in a process called synapsis (si-napsis). Because each chromosome consists of two chromatids, the pairing of the homologous chromosomes brings two chromatids of each chromosome close together, an arrangement called a tetrad (figure 28. Occasionally, part of a chromatid of one homologous chromosome breaks off and is exchanged with part of another chromatid from the other homologous chromosome. This exchange of genetic material between maternal and paternal chromosomes is called crossing over and may result in new gene combinations on the chromosomes. During metaphase I, homologous pairs of chromosomes line up near the center of the cell (figure 28. For each homologous pair, however, the orientation of the maternal and paternal chromosomes is random. The way the chromosomes align during synapsis results in the random assortment of maternal and paternal chromosomes in the daughter cells during meiosis. Crossing over and the random assortment of maternal and paternal chromosomes are responsible for the large degree of diversity in the genetic composition of sperm cells and oocytes produced by each individual. During anaphase I, the homologous pairs are separated to each side of the cell (figure 28. During telophase I, new nuclei form, and the cell completes division of the cytoplasm to form two cells (figure 28. Chromosomes Nucleus Centrioles 2 Middle prophase I Homologous chromosomes synapse to form tetrads. Pair of chromosomes Spindle fibers 3 Metaphase I Homologous chromosomes align at the center of the cell. The cells divide to form four daughter cells with a haploid (n) number of chromosomes. Since the chromosome number is reduced from a 2n number (46 chromosomes, or 23 pairs) to an n number (23 chromosomes, or 1 from each homologous pair) during meiosis I, this division is often called a reduction division. At the end of meiosis I, each of the 23 chromosomes in the daughter cells still consists of two chromatids. Then the chromatids separate at the centromere, and each daughter cell receives one of the chromatids from each chromosome. Consequently, each of the four daughter cells produced by meiosis contains 23 chromosomes. Describe two mechanisms that occur during meiosis that produce genetic variation among gametes. The urethra, in turn, exits the pelvis and passes through the penis to the outside of the body. It is divided into two internal compartments by an incomplete connective tissue septum. Externally, the compartments of the scrotum are marked by a midline irregular ridge called the raphe (rf; a seam). The raphe extends posteriorly to the anus and anteriorly onto the inferior surface of the penis. The wall of the scrotum includes the skin, a layer of superficial fascia consisting of loose connective tissue, and a layer of smooth muscle called the dartos (darts) muscle. In cold temperatures, the dartos muscle contracts, causing the skin of the scrotum to become firm and wrinkled and reducing its overall size. When temperature increases due to a warmer environment or as a result of exercise or fever, the dartos and cremaster muscles relax, and the skin of the scrotum becomes loose and thin, allowing the testes to descend away from the body and keep cool. The response of the dartos and cremaster muscles is important because sperm cells are very temperature-sensitive and do not develop normally if the testes become too warm or too cool. The perineum is divided into two triangles by the superficial transverse muscles and the deep transverse perineal muscles. These muscles run transversely between the two ischial tuberosities (see figure 10. In males, the urogenital (r-jeni-tl) triangle, or anterior triangle, contains the base of the penis and the scrotum, and the smaller anal triangle, or posterior triangle, contains the anal opening (figure 28. Explain the role of the dartos and cremaster muscles in regulating the temperature of the testes. Describe the structure of the testes, the specialized cells of the testes, and the process of spermatogenesis. Describe the structure of the penis, seminal vesicles, prostate gland, and bulbourethral glands and explain their functions. Accessory glands include the seminal vesicles, the prostate gland, and the bulbourethral glands. The testes and epididymides, in which the sperm cells develop, are located outside the body cavity in the scrotum. The ducta deferentia lead from the testes into the pelvis, where they join the ducts of the seminal vesicles to form the ampullae. Extensions of the ampullae, called the ejaculatory ducts, pass into the prostate and empty into the urethra within the prostate. Testes Testicular Histology the testes (testz) are small, oval-shaped organs, each about 45 cm long, within the scrotum (see figure 28. Some structures are drawn as a modified medial section to reveal the testis, epididymis, and seminal vesicles and to show the relationship of the ductus deferens to the ureter and urinary bladder. The combined length of the seminiferous tubules in both testes is nearly half a mile. Considering that sperm cells are produced in the seminiferous tubules, it is not surprising that adult males are capable of producing such high numbers of gametes. The seminiferous tubules empty into a set of short, straight tubules called the tubuli recti. The rete testis empties into 1520 tubules called efferent ductules (dktools) that pass through the tunica albuginea to exit the testis. The efferent ductules have a ciliated pseudostratified columnar epithelium, which helps move sperm cells out of the testis. Descent of the Testes secretion is sperm cells, and their major endocrine secretion is the hormone testosterone. The outer part of each testis is a thick, white capsule consisting mostly of fibrous connective tissue called the tunica albuginea (al-b-jin-). Extensions of the tunica albuginea extend into the testis and form incomplete septa (sept; figure 28. The lobules contain seminiferous (semi-nifer-s; seed carriers) tubules, in which sperm cells develop. Loose connective tissue surrounding 1034 By approximately 8 weeks following fertilization, the testes have developed as retroperitoneal organs. Initially, the testes are located high in the abdominopelvic cavity near the developing kidneys. Each testis is connected to a labioscrotal swelling by a gubernaculum (goober-nak-lm), a fibromuscular cord (figure 28. The testes descend toward the area where the inguinal (inggwi-nl) canals will form (figure 28. Spermatogonia are near the periphery, and mature sperm cells are near the lumen of the seminiferous tubules. Approximately 12 weeks 2 the testis beneath the parietal peritoneum is descending toward the inguinal canal. Inguinal canal 3 Process vaginalis Testis Urinary bladder Ductus deferens Inguinal canal Process vaginalis (degenerated) Tunica vaginalis 4 Urinary bladder Ductus deferens Testis Scrotal ligament Scrotum Adult Gubernaculum Scrotum Between 7 and 9 months 3 the testis follows the process vaginalis and descends through the inguinal canal into the scrotum. Between 7 and 9 months of development, the testes move through the inguinal canals into the scrotum (figure 28. As it moves into the scrotum, each testis is preceded by an outpocketing of the peritoneum called the process vaginalis (vajin-lis). The superior part of each process vaginalis usually degenerates, and the inferior part remains as a small, closed sac called the tunica (tooni-k) vaginalis (figure 28. The tunica vaginalis is a serous membrane consisting of a layer of simple squamous epithelium resting on a basement membrane. The tunica vaginalis surrounds most of the testis in much the same way that the pericardium surrounds the heart. The tunica vaginalis secretes a small amount of fluid, which allows the testes to move in the scrotum, producing little friction. The testes have descended into the scrotum in approximately 79% of male infants delivered prior to 28 weeks of development. In male infants delivered after 28 weeks, greater than 97% show normal testes descent. The inguinal canals are bilateral, oblique passageways in the anterior abdominal wall. They originate at the deep inguinal rings, which open through the aponeuroses of the transversus abdominis muscles. The canals extend inferiorly and obliquely and end at the superficial inguinal rings, openings in the aponeuroses of the external abdominal oblique muscles. In females, the inguinal canals develop, but they are much smaller than in males, and the ovaries do not normally descend through them. Because the higher temperature of the abdominal cavity prevents normal sperm cell development, sterility can result if both testes are involved (see chapter 29). In addition, approximately 10% of testicular cancer cases occur in men with a history of cryptorchidism. List the ducts that will move sperm from the seminiferous tubules out of the testis. Sperm Cell Development Before puberty, the testes remain relatively simple and unchanged from the time of their initial development. The interstitial cells are not particularly prominent during this period, and the seminiferous tubules lack a lumen and are not yet functional. At 1214 years of age, the interstitial cells increase in number and size, a lumen develops in each seminiferous tubule, and sperm cell production begins. Normally, the inguinal canals are closed, but they represent weak spots in the abdominal wall. If the deep inguinal ring remains open, or if it is weak and enlarges later in life, a loop of intestine can protrude into or even pass through the inguinal canal, resulting in an indirect inguinal hernia. A direct inguinal hernia results from a tear, or rupture, in a weakened area of the anterior abdominal wall near the inguinal canal, but not through the inguinal canal. Inguinal hernias can be quite painful and even very dangerous, especially if a portion of the small intestine is compressed so that its blood supply is cut off. A Spermatogenesis (sperm-t-jen-sis), sperm cell development, occurs in the seminiferous tubules (figure 28. The seminiferous tubules contain two types of cells, germ cells and sustentacular (ss-ten-tak-lr) cells, or Sertoli (sr-tl) cells (also sometimes referred to as nurse cells). The germ cells are the ones that divide and differentiate during spermatogenesis to form sperm cells. The sustentacular cells are large cells that extend from the periphery to the lumen of the seminiferous tubule (figure 28. The sustentacular cells nourish the germ cells and probably produce, together with the interstitial cells, a number of hormones, such as androgens, estrogens, and inhibins. In addition, tight junctions between the sustentacular cells form a blood-testis barrier between germ cells and sperm cells. The blood-testis barrier isolates the sperm cells from the immune system (figure 28. This barrier is necessary because, as the sperm cells develop, they form surface antigens that could stimulate an immune response, resulting in their destruction. Interstitial cells secrete testosterone, which passes into the sustentacular cells and binds to intracellular receptors. In addition, testosterone in the sustentacular cells is converted to two other steroids: dihydrotestosterone (d-hdr-tes-toster-n) and estradiol, a specific type of estrogen. The sustentacular cells also secrete a protein called androgen-binding (andr-jen) protein into the seminiferous tubules. Testosterone and dihydrotestosterone bind to androgenbinding protein and are carried along with other secretions of the seminiferous tubules to the epididymis. Estradiol and dihydrotestosterone may be the active hormones that promote sperm cell formation. The most peripheral cells, those adjacent to the basement membrane of the seminiferous tubules, are spermatogonia (sperm-t-gn-), which divide by mitosis (figure 28.
The lymphatic trunks either connect to large veins in the thorax or join to yet larger vessels called lymphatic ducts medicine and science in sports and exercise septra 480 mg fast delivery, which then connect to the large veins symptoms 0f a mini stroke buy septra cheap online. Many connect at the junction of the internal jugular and subclavian veins symptoms mono cheap 480 mg septra otc, but connections on the subclavian medications used to treat anxiety septra 480mg order, the jugular treatment 5th finger fracture purchase 480mg septra with amex, and even the brachiocephalic veins exist. On the right side, the jugular, subclavian, and bronchomediastinal trunks typically join a right thoracic vein separately (figure 22. However, in about 20% of people, the three trunks converge to form a single, short duct 1 cm in length called the right lymphatic duct (see figure 22. These trunks drain lymph from the right side of the head, upper-right limb, and right thorax (figure 22. It is approximately 3845 cm in length, extending from the twelfth thoracic vertebra to the base of the neck (figure 22. The thoracic duct drains lymph from the right side of the body inferior to the thorax and the entire left side of the body (figure 22. The bronchomediastinal trunk sometimes connects to the thoracic duct but typically joins a vein. The intestinal and lumbar trunks, which drain lymph inferior to the diaphragm, supply the inferior end of the thoracic duct. They can directly join the thoracic duct or merge to form a network that connects to the thoracic duct. In a small proportion of cases, the lymphatic trunks form a sac called the cisterna chyli (sis-tern kl). In summary, excess interstitial fluid enters lymphatic capillaries, forming lymph. Lymph passes through the lymphatic vessels and through associated lymph nodes, where it is filtered. Lymphatic vessels converge to form larger lymphatic trunks, which drain lymph from major regions of the body. Lymphatic trunks empty directly into thoracic veins or combine to form larger lymphatic ducts, which empty into thoracic veins. What areas of the body are drained by the right lymphatic trunks, left lymphatic trunks, and thoracic duct Lymphatic Tissue and Organs Lymphatic organs contain lymphatic tissue, which consists primarily of lymphocytes but also includes macrophages, dendritic cells, reticular cells, and other cell types. The two types of lymphocytes, B cells and T cells, originate from red bone marrow and are carried by the blood to lymphatic organs and other tissues. When the body is exposed to microorganisms or other foreign substances, the lymphocytes divide, increase in number, and become part of the immune response that destroys microorganisms and foreign substances. In addition to cells, lymphatic tissue is composed of very fine collagen fibers, called reticular fibers produced by a specific type of fibroblast called a reticular cell. When lymph or blood passes through lymphatic organs, the fiber network traps microorganisms and other particles, filtering the fluid. Lymphatic tissue of the body may or may not be separated from other tissues by a connective tissue capsule. Lymphatic tissue that is not surrounded by a connective tissue capsule is called nonencapsulated, whereas lymphatic tissue surrounded by a capsule is said to be encapsulated. Encapsulated lymphatic tissue forms most of the organs of the lymphatic tissue: lymph nodes, the spleen, and the thymus. Aggregations of nonencapsulated lymphatic tissue are often associated with organs of other systems. In these locations, the lymphatic tissue is well positioned to intercept microorganisms as they enter the body. Diffuse Lymphatic Tissue and Lymphatic Nodules Diffuse lymphatic tissue contains dispersed lymphocytes, macrophages, and other cells. Diffuse lymphatic tissue has no clear boundary and blends with surrounding tissues (figure 22. Lymphatic nodules are denser arrangements of lymphatic tissue organized into compact, somewhat spherical structures ranging in size from a few hundred microns to a few millimeters or more in diameter (figure 22. Lymphatic nodules are numerous in the loose connective tissue of the digestive, respiratory, urinary, and reproductive systems. Peyer patches are aggregations of lymphatic nodules in the distal half of the small intestine and the appendix. Distinguish among lymphatic tissue, lymphatic nodules, Peyer patches, and lymphatic follicles. Lymph Nodes Lymph nodes are small, round or bean-shaped structures ranging from 1 mm to 25 mm long. An important function of lymph nodes is to filter the lymph, removing bacteria and other materials. In addition, lymphocytes congregate, function, and proliferate within lymph nodes. Superficial lymph nodes are in the subcutaneous tissue beneath the skin, and deep lymph nodes are everywhere else. Cervical and head lymph nodes (about 70) filter lymph from the head and neck, axillary lymph nodes (about 30) filter lymph from the upper limbs and superficial thorax, thoracic lymph nodes (about 100) filter lymph from the thoracic wall and organs, abdominopelvic lymph nodes (about 230) filter lymph from the abdomen and pelvis, and inguinal and popliteal lymph nodes (about 20) filter lymph from the lower limbs and the superficial pelvis. Extensions of the capsule, called trabeculae (trbek-l), form a delicate internal skeleton in the lymph node. Reticular fibers extend from the capsule and trabeculae to form a fibrous network throughout the lymph node. In some areas of the lymph node, lymphocytes and macrophages are packed around the reticular fibers to form lymphatic tissue; in other areas, the reticular fibers extend across open spaces to form lymphatic sinuses. The lymphatic tissue and sinuses within the lymph node are arranged into two somewhat indistinct layers, an outer cortex and an inner medulla. The cortex consists of a subcapsular sinus, beneath the capsule, and cortical sinuses, which are separated by diffuse lymphatic tissue, trabeculae, and lymphatic nodules. The inner medulla is organized into branching, irregular strands of diffuse lymphatic tissue, called the medullary cords, separated by medullary sinuses. Afferent lymphatic vessels carry lymph to the lymph nodes, where it is filtered, and efferent lymphatic vessels carry lymph away from the lymph nodes. Lymph from afferent lymphatic vessels enters the subcapsular sinus, filters through the cortex and medulla, and exits the lymph node through efferent lymphatic vessels. The efferent vessels of one lymph node may become the afferent vessels of another lymph node or may converge to form lymphatic trunks, which carry lymph to the blood at thoracic veins. Macrophages lining the lymphatic sinuses remove bacteria and other foreign substances from the lymph as it slowly filters through the sinuses. Microorganisms and other foreign substances Tonsils Tonsils are large groups of lymphatic nodules and diffuse lymphatic tissue located deep to the mucous membranes within the pharynx (throat; figure 22. The tonsils protect against bacteria and other potentially harmful material entering the pharynx from the nasal or oral cavity. Though we often just think of one pair of tonsils, there are actually three groups of tonsils: (1) the palatine tonsils, (2) the pharyngeal tonsil, and (3) the lingual tonsil. They are relatively large, oval, lymphatic masses on each side of the junction between the oral cavity and the pharynx. The pharyngeal (f-rinj-l) tonsil is a collection of somewhat closely aggregated lymphatic nodules near the junction between the nasal cavity and the pharynx. When the pharyngeal tonsil is enlarged, it is commonly called the adenoid (ad-noyd; glandlike) or adenoids (ad-noydz). The lingual tonsil is a loosely associated collection of lymphatic nodules on the posterior surface of the tongue. Sometimes the palatine or pharyngeal tonsils become chronically infected and must be removed. The lingual tonsil becomes infected less often than the other tonsils and is more difficult to remove. The newly produced lymphocytes are released into the lymph and eventually reach the bloodstream, where they circulate. Subsequently, the lymphocytes can leave the blood and enter other lymphatic tissues. Spleen the spleen is roughly the size of a clenched fist and is located on the left, superior part of the abdominal cavity (figure 22. The size and weight of the spleen tend to decrease in older people, but in certain diseases the spleen can achieve a weight of 2000 g or more. For example, about 50% of individuals who have infectious mononucleosis develop an enlarged spleen as a result of increased numbers of defense cells. The spleen has an outer capsule of dense irregular connective tissue and a small amount of smooth muscle. Bundles of connective tissue fibers from the capsule form trabeculae, which extend into the organ, subdividing it into small, interconnected compartments. Arteries, veins, and lymphatic vessels extend through the trabeculae to supply the compartments, which are filled with white and red pulp. Red pulp consists of a fibrous network, filled with macrophages and red blood cells, and enlarged capillaries that connect to the veins. Branch of trabecular artery 1 Periarterial lymphatic sheath Lymphatic nodule 2 Venous sinus Splenic cord 3 A few capillaries directly connect to a venous sinus. White pulp 4 the ends of most capillaries are separated from the beginning of the venous sinuses by a small gap. Blood percolates through the splenic cords and Capillaries passes through the walls of the venous sinuses. Branches of the splenic (splenik) artery enter the spleen at the hilum, and their branches follow the various trabeculae into the spleen (figure 22. From the trabeculae, arterial branches extend into the white pulp, which consists of the periarterial lymphatic sheath and lymphatic nodules (figure 22. The periarterial lymphatic sheath is composed of diffuse lymphatic tissue surrounding arteries and arterioles extending to lymphatic nodules. Blood flows from the capillaries into the red pulp, which consists of the splenic cords and the venous sinuses. The splenic cords are a network of reticular cells that produce reticular fibers (see chapter 4). The spaces between the reticular cells are occupied by splenic macrophages and blood cells that have come from the capillaries. The venous sinuses typically connect to trabecular veins, which unite to form vessels that leave the spleen to form the splenic vein. Before removing the cancerous tumor from her left breast, her surgeon injected a dye and a radioactive tracer, technetium-99, at the tumor site. The dye and tracer enabled the surgeon to find sentinel lymph nodes, those lymph nodes closest to the tumor. A few days after the surgery, Cindy noticed that the skin on her left arm felt tight and the arm felt heavy. In the United States, the most common cause of lymphedema is removal of or damage to lymph nodes and vessels as a result of cancer surgery or radiation treatment. Approximately 1020% of women whose axillary lymph nodes have been removed develop lymphedema. There are high concentrations of T cells in the periarterial lymphatic sheath and B cells in the lymphatic nodules. The resulting small increase in circulating red blood cells can promote better oxygen delivery to muscles during exercise or emergency situations. Physiologists do not presently know if this reduction results from the contraction of smooth muscle within the capsule, from the contraction of smooth muscle (myofibroblast) within the trabeculae, or from the reduced blood flow through the spleen caused by the constriction of blood vessels. Although the ribs protect the spleen, it is often ruptured in traumatic abdominal injuries. If these techniques do not stop the bleeding, a splenectomy (spl-nekt-m), removal of the spleen, may be necessary. After removal of the spleen, other lymphatic organs and the liver compensate for the loss of its functions. In intermittent pneumatic pump compression therapy, the pressure of a garment enclosing a limb increases and decreases periodically. In addition, the pressure increases sequentially from the distal part to the proximal part of a limb. The thymus increases in size until the first year of life, after which it remains approximately the same size until 60 years of age, when it decreases in size (see section 22. Trabeculae extend from the capsule into the substance of the gland, dividing it into lobules. Unlike other lymphatic tissue, which has a fibrous network of reticular fibers, the framework of thymic tissue consists of epithelial cells. The processes of the epithelial cells are joined by desmosomes, and the cells form small, irregularly shaped compartments filled with lymphocytes. Near the capsule and trabeculae, the lymphocytes are numerous and form dark-staining areas of the lobules called the cortex. A lighter-staining, central portion of the lobules, called the medulla, has fewer lymphocytes. The medulla also contains rounded epithelial structures, called thymic corpuscles (Hassall corpuscles). For more than 150 years, the function of these structures was unknown, but current research indicates that thymic corpuscles function in the development of regulatory T cells. Thymosin, a hormone secreted by the thymus, is important in the T-cell maturation process. The lymphocytes that survive the maturation process are capable of reacting to foreign substances, but they normally do not react to and destroy healthy body cells (see "Origin and Development of Lymphocytes," in section 22. These surviving thymic lymphocytes migrate to the medulla, enter the blood, and travel to other lymphatic tissues. Although blood flow through the spleen can take from a few seconds to an hour or more, most blood flows through the spleen rapidly. The rapid flow results from the movement of blood from the ends of capillaries into the beginning of the venous sinuses.
Buy cheap septra 480mg on line. டெஙà¯à®•à¯ காயà¯à®šà¯à®šà®²à¯ | Dengue fever Symptoms treatments prevention Dengue Awareness |Dengue tamil tips.

So medications 2 purchase septra line, if the lateral rectus is weak and does not pull the eye laterally treatment jock itch generic 480 mg septra with mastercard, the medial rectus will predominate world medicine cheap generic septra uk, and the eye will deviate medially medicine keri hilson lyrics order septra 480mg with mastercard. Next symptoms e coli discount septra 480 mg otc, it is necessary to know the location of the supraspinatus muscle to answer the question. There, you see that in extreme abduction the supraspinatus would be compressed against the acromion process of the scapula. When doing chin-ups the elbow flexes as you pull your body up toward the bar and extends as you lower your body back down. Notice that the biceps brachii both flexes the elbow and supinates the forearm; however, the brachialis only flexes the elbow. When the forearm is pronated, the biceps brachii cannot contract as forcefully when flexing the elbow because the distal end of the biceps brachii is partially medially rotated. When the forearm is supinated (palms toward you), both muscles can work optimally, making these the easier chin-ups. At this point in the reading, we know that axons are a cellular projection from the neuron, sometimes called nerve fibers. We also know that the cell body is the site of protein synthesis for the entire cell. Therefore, if the distal portion of an axon is severed from the rest of the cell, it will die. There is no way for the distal axon to replenish the enzymes and other proteins essential for survival. On the other hand, any remaining portion of axon still attached to the cell body survives and, in many cases, grows to replace the severed portion. Although this question seems to ask about the cause of death, it is really asking about what cellular changes nervous tissue would exhibit in the event of trauma. First recall the types of glial cells found in central nervous tissue, in particular any cells involved in tissue repair or protection. They are defined as "cells that become mobile and phagocytic in response to inflammation" and are described as cells that "migrate to areas damaged by infection, trauma, or stroke. The pathologist recognized the prior stroke because of the large number of microglia in the brain tissue surrounding the blocked blood vessels. In addition, the pathologist knew at least one damaged area was several weeks old because it takes time for microglia to migrate to a damaged area. However, the second damaged area of the brain showed evidence of a reduced blood supply, but far fewer microglia. Recall from chapter 4 that cardinal signs of inflammation include redness and edema, both of which were evident in the area of the brain injured by the fall. Therefore, the pathologist could conclude that either the second stroke killed the man, and caused him to fall, or it caused him to fall and the trauma to the brain incurred during the fall caused his death. The first step to answer this question is to define the basis for the resting membrane potential. Recall that the inside of the plasma membrane is more negative than the outside of the plasma membrane due to a higher concentration of negatively charged proteins inside the cell. The K+ is also in higher concentration inside the cell and tends to "offset" the negative charge of the proteins. However, the K+ leak channels allow K+ to diffuse out of the cell, down the concentration gradient. Removal of K+ removes positive charge inside the cell, and the membrane potential becomes more negative. Therefore, because Tissue A has more K+ leak channels, more K+ can leak out of Tissue A cells than in Tissue B cells, and the inside of Tissue A cells becomes more negative. The resting membrane potential is larger for Tissue A than for Tissue B; it is further from threshold. First, consider the role Ca2+ plays in establishing the resting membrane potential. We learned that Ca2+ ions are important for keeping voltage-gated Na+ channels closed. When voltage-gated Na+ channels open, the cell depolarizes, or becomes more positively charged on the inside of the plasma membrane. Thus, if Ca2+ levels drop, the voltage-gated Na+ channels open, Na+ diffuses into the cell, making it more positive, and the resting membrane potential increases. To answer this question, let us first describe the Na+ concentration and its role in a normal excitable cell. We learned that, in a normal cell, the concentration of Na+ is much higher outside the cell than inside the cell. As a result, when Na+ channels open, Na+ diffuses into the cell quickly, causing the changes in the membrane potential that result in an action potential. The movement of Na+ into the cell is the result of its steep concentration gradient. Remember, also, that enough Na+ must enter a cell for the membrane potential to reach threshold, opening the voltage-regulated Na+ channels and causing an action potential. If the extracellular concentration of Na+ were reduced, then the concentration gradient would also be reduced. The effect would be that, if the cell were stimulated, less Na+ would enter the cell. Threshold is the membrane potential at which voltage-gated Na+ channels first begin to open. A prolonged threshold stimulus will result in a single action potential at the end of each relative refractory period. However, a prolonged, stronger-than-threshold stimulus will produce a greater number of action potentials than a threshold stimulus of the same duration due to a higher Na+ permeability in the axon. The stronger-than-threshold stimulus will even produce action potentials during the relative refractory period. Therefore, a neuron receiving a stronger stimulus will send a greater frequency of action potentials. Because of the all-or-none properties of neurons, one action potential has the same magnitude as another. Recall that it is the time during which a neuron cannot fire an action potential no matter how strong the stimulus. Thus, if the axon is nonresponsive for 1 ms, action potentials can be generated no faster than every millisecond. Because there are 1000 ms in 1 second, the maximal frequency is 1000 action potentials/second. The question asks us to compare signal transmission rates of electrical and chemical synapses. Chemical synapses rely on diffusion of the neurotransmitter across the synaptic cleft, binding to the receptor and then triggering a signal transduction pathway. Thus, electrical synapses take no more time to be transmitted than an individual action potential takes traveling from node to node in a myelinated axon, or down the axon in an unmyelinated axon. On the other hand, the chemical synapse is more complex and reliant on molecular conformation change, enzyme activity, and so on, all of which are slower processes. Neuron C does not require as much time for temporal summation to reach threshold to fire an action potential when stimulated by neuron B vs. Therefore, neuron C fires more action potentials with stimulation by neuron B alone than with temporal summation of neuron A alone. In other words, the neurotransmitter from neuron B is more effective when released with the neuromodulator from neuron B than it is when released alone from neuron A. Similarly, in this chapter we learned that the dorsal root ganglia are clusters of sensory cell bodies and the dorsal roots are bundles of sensory axons. The ganglia are larger in diameter than the roots because of the size difference between the cell bodies and axons. Recall that spinal nerves have both sensory and motor axons, so action potentials are propagated both to the spinal cord and away from the spinal cord. Dorsal roots contain only sensory axons, so action potentials are conducted to the spinal cord only. Finally, ventral roots contain only motor axons, so action potentials are conducted away from the spinal cord. To answer this question, look at the left arm, forearm, and hand to see which nerves are indicated. C5 and T1 innervate the left arm, C6 and T1 innervate the forearm, and C7 and C8 innervate the hand. Recall that the phrenic nerve innervates the diaphragm, allowing for the contraction necessary for breathing. If the right phrenic nerve were damaged, then we would expect lack of muscle contraction in the right half of the diaphragm, affecting breathing. To answer the second part of the question, we need to consider the location of spinal cord injury to predict the effect it would have on the diaphragm. Remember that the phrenic nerve is part of the cervical plexus, which includes spinal nerves C1C4. On the other hand, if the spinal cord were completely severed at the level of C6, the phrenic nerve would not be damaged, and the diaphragm would not be affected. Also after reviewing the description of the brachial plexus and viewing figures 12. An x-ray of the neck can indicate the presence of extra rib, which most commonly affects the roots C8T1. Based on our answers for parts a and b, we can assume that the ulnar nerve is most likely affected, so the muscles innervated by the ulnar nerve may be affected. However, the radial and median nerves are also associated with the C8T1 root and may be affected; therefore, the muscles innervated by these might also be affected. The sources of nerve fibers in the femoral nerve are L2, L3, and L4; therefore, the intervertebral disk involved compresses L2, L3, or L4 on the left side of the vertebral column. We can see from the map that L3 innervates the dermatome of the medial thigh and is the most likely spinal nerve involved. The referred pain results from compression of the spinal nerve that innervates the medial thigh and the knee. The compression stimulates action potentials in the nerves, and the pain is referred to the site of the sensory receptors for that nerve. We learned in this chapter that reflexes that maintain blood pressure are integrated by the medulla oblongata. Similarly, the reflexes cause the constriction of blood vessels in the skin and viscera to increase blood volume and therefore blood pressure. Recall that respiratory reflexes are integrated in the medulla oblongata and the pons. The Clinical Impact, "Traumatic Brain Injuries and Hematomas," explains that, often, when one part of the head suffers a heavy blow, the brain moves within A-35 Appendix G the cranial cavity and hits the opposite side of the cranial cavity. In this case, the blow to the back of the head forced the brain anteriorly, and the frontal lobes struck the frontal bones with enough force to tear blood vessels between the brain and the dura. Subsequent bleeding from these vessels into the subdural space created the subdural hematoma. This type of injury is a countercoup brain injury because it occurs on the side of the brain opposite the point of traumatic impact. We can assume that the herniation of the medulla into the vertebral canal compressed the tissue of the medulla and therefore disrupted its function. Recall from its description that the medulla oblongata contains the centers for controlling respiration and heart rate. To answer this question, let us first review the functions of the oculomotor, trochlear, and abducens nerves, listed in table 13. Besides innervating the levator palpebrae superioris muscle, the oculomotor nerve innervates the four eye muscles that move the eyeball so that the gaze is directed superiorly, inferiorly, medially, or superolaterally. If the patient can move the eyes in these directions, the oculomotor nerve is not damaged. Similarly, the abducens nerve directs the gaze laterally, and the trochlear nerve directs the gaze inferolaterally. If the patient can move the eyes in these directions, the associated nerves are intact. To answer this question we must first determine the function of the sternocleidomastoid muscle. If the innervation to one sternocleidomastoid muscle is eliminated due to accessory nerve injury, the opposite muscle is unopposed and turns the face toward the side of the injury. A person with wry neck whose head is turned to the left most likely has an injured left accessory nerve. From this we can conclude that a lesion on one side of the spinal cord that interrupts the spinothalamic tract would eliminate the specific sensations carried by that tract below the level of the lesion, but on the opposite side of the body. Pain and temperature sensation from the opposite side of the body below the lesion would be eliminated. There would be few, if any, clinical changes in detecting light touch because other tracts still carry this information. We can also specifically identify the fasciculus gracilis tract, since Bill and Mary lost the sensations in the lower half of the body. The next step is to determine why Bill and Mary have similar symptoms but different injuries. Recall that the secondary neuron axons cross over along the sensory pathway, so that stimuli from one side of the body are conveyed to the primary sensory cortex on the opposite side. We can see that the crossover of axons for the fasciculus gracilis tract occurs in the medulla, which is part of the brainstem. Since the axons have not crossed over in the spinal cord, we can assume that the spinal cord injury was on the left side, the same side as the sensory receptors.
On the upper limb medications during pregnancy buy cheap septra 480mg on line, a pulse can also be felt in three arteries: (1) the axillary artery in the axilla medicine 3 times a day purchase septra toronto, (2) the brachial artery on the medial side of the arm slightly proximal to the elbow medicine 100 years ago cheap septra 480 mg buy line, and (3) the radial artery on the lateral side of the anterior forearm just proximal to the wrist symptoms 20 weeks pregnant buy septra uk. The radial pulse medicine for nausea order septra overnight, taken at the radial artery, is traditionally used because it is the most easily accessible artery in the body. In the lower part of the body, a pulse can be felt in four locations: (1) the femoral artery in the groin, (2) the popliteal artery just proximal to the knee, (3) the dorsalis pedis artery at the ankle, and (4) the posterior tibial artery at the ankle. As the pulse passes through the smallest arteries and arterioles, it is gradually damped, so that the fluctuation between the systolic Superficial temporal artery Common carotid artery Facial artery Axillary artery Brachial artery Radial artery and diastolic pressures becomes smaller until the difference is almost absent at the end of the arterioles (see figure 21. At the beginning of the capillary, there is a steady pressure of close to 30 mm Hg, which is adequate to force blood through the capillaries if the precapillary sphincters dilate. Capillary Exchange and Regulation of Interstitial Fluid Volume Approximately 10 billion capillaries exist in the body. The heart and blood vessels maintain blood flow through those capillaries and support capillary exchange, which is the movement of substances into and out of capillaries. Capillary exchange is the process by which cells receive everything they need to survive and to eliminate metabolic waste products. Oxygen, hormones, and nutrients, such as glucose and amino acids, diffuse from a higher concentration in capillaries to a lower concentration in the interstitial fluid. Waste products, including carbon dioxide, diffuse from a higher concentration in the interstitial fluid to a lower concentration in the capillaries. Lipid-soluble molecules, such as oxygen, carbon dioxide, steroid-hormones, and fatty acids, diffuse through the plasma membranes of the endothelial cells of the capillaries. Watersoluble substances, such as glucose and amino acids, diffuse through intercellular spaces or through fenestrations of capillaries. In a few areas of the body, such as the spleen and liver, the spaces between the endothelial cells are large enough to allow proteins to pass through them. In other areas, the connections between endothelial cells are extensive, and few molecules pass between the endothelial cells; such is the case in the capillaries of the brain that form the blood-brain barrier. In these capillaries, mediated transport moves water-soluble substances across the capillary walls (see chapter 13 for a description of the blood-brain barrier). The endothelial cells of capillaries appear to take up small pinocytotic vesicles and transport them across the capillary wall. However, the pinocytotic vesicles do not appear to be a major means by which molecules move across the capillary wall. Blood enters capillaries at their arterial ends and exits the capillaries at their venous ends. A small amount of fluid moves out of capillaries at their arterial ends, and most of that fluid reenters the capillaries at their venous ends (figure 21. The remaining fluid enters lymphatic vessels, which eventually return it to the venous circulation (see chapter 22). Alterations in the forces affecting fluid movement across capillary walls are responsible for edema (see Clinical Impact, "Edema and Capillary Exchange"). Net hydrostatic pressure is the difference in pressure between the blood and the interstitial fluid. When the net osmotic pressure is subtracted from the net hydrostatic pressure, the result is a positive net filtration pressure that causes fluid to move out of the capillary. When the net osmotic pressure is subtracted from the net hydrostatic pressure, the result is a negative net filtration pressure that causes fluid to move into the capillary. This pressure results mainly from the force of contraction of the heart, but it can be modified by the effect of gravity on fluids within the body (see "Blood Pressure and the Effect of Gravity," later in this section). Here, it is only necessary to understand that excess interstitial fluid enters lymphatic capillaries and is eventually returned to the blood. Net osmotic pressure is the difference in osmotic pressure between the blood and the interstitial fluid. Osmotic pressure is the result of solute differences across a selectively permeable barrier, such as a capillary wall (see chapter 3). Solutes, such as proteins in the blood and interstitial fluids, will greatly affect the osmotic pressure. Large proteins do not pass freely through the capillary walls, and the difference in protein concentrations between the blood and the interstitial fluid is responsible for osmosis across the capillary wall. Ions and small molecules do not make a significant contribution to osmosis across the capillary wall because they pass freely through it and their concentrations are approximately the same in the blood as in the interstitial fluid. This causes an increase in the interstitial colloid osmotic pressure, which causes a net increase in the amount of fluid moving from capillaries into interstitial spaces. Chemical mediators of inflammation increase the permeability of the capillary walls and can cause edema. Decreases in plasma protein concentration reduce the blood colloid osmotic pressure, so more fluid moves out of the capillary at its arterial end and less fluid moves into the capillary at its venous end. Severe liver infections that reduce plasma protein synthesis, loss of protein molecules in urine through the kidneys, and protein starvation all lead to edema. Blockage of veins, as in venous thrombosis, increases blood pressure in capillaries and can cause edema. Either blockage or removal of lymphatic vessels, as occurs when lymph nodes are suspected of being cancerous, allows fluid to accumulate in the interstitial spaces and results in edema. The concentration of proteins within capillaries and the concentration of proteins within interstitial fluid do not change significantly because only a small amount of fluid passes from the capillaries into the tissue spaces. Therefore, the net osmotic pressure moving fluid into capillaries by osmosis is still approximately 20 mm Hg. Disruptions in the movement of fluid across the wall of the capillary can result in edema, or swelling, as a result of increased interstitial fluid volume. Predict 5 Edema often results from a disruption in the normal inwardly and outwardly directed pressures across the capillary wall. On the basis of what you know about fluid movement across the wall of the capillary and the regulation of capillary blood pressure, explain why large fluctuations in arterial blood pressure do not cause significant edema, whereas small increases in venous pressure can lead to edema. Describe the factors that influence the movement of fluid from capillaries into the tissues. The greater the osmotic pressure of a fluid, the greater the tendency for water to move into that fluid (see chapter 3). The net osmotic pressure results in the osmosis of water into the capillary because water has a greater tendency to move into the blood than into the interstitial fluid. The net filtration pressure at the arterial end of the capillary is equal to the net hydrostatic pressure, which moves fluid out of the capillary, minus the net osmotic pressure, which moves fluid into the capillary. One important factor that is influenced by veins is the preload, which is determined by the volume of blood that enters the heart from the veins (see chapter 20). Therefore, the factors that affect flow in the veins are of great importance to the overall function of the cardiovascular system. If the volume of blood is increased because of a rapid transfusion, the amount of blood flow to the heart through the veins increases. This increases the preload, which causes the cardiac output to increase because of the Starling the blood pressure decreases as blood moves through the capillary. The decrease is from about 30 mm Hg at the arterial end of the capillary to 10 mm Hg at the venous end of the capillary. One night, as he walked to his car after class, Harry noticed that his right leg was uncomfortable. When he arrived home, about 90 minutes later, Harry realized that the calf of his right leg had become very swollen. When he extended his knee and plantar flexed his foot, the pain in his right leg increased. In the emergency room, technicians performed a Doppler test, which monitors the flow of blood through blood vessels. Harry remained in the hospital for several days, during which heparin was continued and then oral coumadin was prescribed. In addition, Harry was required to have his prothrombine time checked periodically. If a thrombus in the posterior tibial vein gave rise to an embolus, name in order the parts of the circulatory system the embolus would pass through before lodging in a blood vessel in the lungs. On the other hand, rapid loss of a large volume of blood decreases venous return to the heart, which decreases the preload and cardiac output. Venous tone is a continual state of partial contraction of the veins as a result of sympathetic stimulation (see chapter 16). Increased sympathetic stimulation increases venous tone by causing the veins to constrict more, which forces the large venous volume to flow toward the heart. Consequently, venous return and preload increase, causing an increase in cardiac output. Conversely, decreased sympathetic stimulation decreases venous tone, allowing veins to relax and dilate. As the veins fill with blood, venous return to the heart, preload, and cardiac output decrease. The periodic muscular compression of veins forces blood to flow more rapidly through them toward the heart. The valves in the veins prevent flow away from the heart, so that, when veins are compressed, blood is forced toward the heart. The combination of arterial dilation and compression of the veins by muscles during exercise causes blood to return to the heart more rapidly than under conditions of rest. Blood Pressure and the Effect of Gravity Blood pressure is approximately 0 mm Hg in the right atrium and approximately 100 mm Hg in the aorta. However, the pressure in the vessels above and below the heart is affected by gravity. While a person is standing, the pressure in the venules of the feet can be as much as 90 mm Hg, instead of the usual 10 mm Hg. Arterial pressure is influenced by gravity to the same degree; thus, the arterial ends of the capillaries can have a pressure of 110 mm Hg rather than 30 mm Hg. The normal pressure difference between the arterial and the venous ends of capillaries remains the same, so that blood continues to flow through the capillaries. The major effect of the high pressure in the feet and legs when a person stands for a prolonged time without moving is edema. Without muscular movement, the pressure at the venous end of the capillaries increases. Up to 1520% of the total blood volume can pass through the walls of the capillaries into the interstitial spaces of the lower limbs during 15 minutes of standing still. When a person changes position from lying down to standing, the blood pressure in the veins of the lower limbs increases. Because of the structure of their walls, the compliance of veins is approximately 24 times greater than the compliance of arteries. The increased blood pressure causes the distensible (compliant) veins to expand but has little effect on the arteries. As the veins expand and fill with blood, venous return decreases because less blood is returning to the heart. As venous return decreases, cardiac output and blood pressure decrease (see chapter 20). If negative-feedback mechanisms do not compensate and cause blood pressure to increase, the delivery of blood to the brain is not adequate to maintain homeostasis, and the person may feel dizzy or even faint. What effect does standing still for a prolonged time have on the blood pressure in the feet and in the head Why does a person feel dizzy if he or she stands up too quickly from sitting or lying down Blood flow provided to the tissues by the circulatory system is highly controlled and matched closely to the metabolic needs of tissues. Mechanisms that control blood flow through tissues are classified as (1) local control and (2) nervous and hormonal control (table 21. Local Control of Blood Flow in Tissues Blood flow is not equal in all tissues of the body. By contrast, blood flow through resting skeletal muscles is not high, but it is greater than that through other tissue types because skeletal muscle constitutes 3540% of the total body mass. However, blood flow through exercising skeletal muscles can increase up to 20-fold, and the flow through the viscera, including the kidneys and liver, either remains the same or decreases. Local control of blood flow is achieved by the periodic relaxation and contraction of precapillary sphincters regulating blood flow through the tissues. In some tissues, blood flow serves purposes other than delivering nutrients and removing waste products. In the kidneys, it eliminates metabolic waste products, regulates water balance, and controls the pH of body fluids. Among other functions, blood flow delivers nutrients that enter the blood from the small intestine to the liver for processing. Functional Characteristics of the Capillary Bed the innervation of the metarterioles and the precapillary sphincters in capillary beds is sparse. As the rate of metabolism increases in a tissue, blood flow through its capillaries increases. The precapillary sphincters relax, allowing blood to flow into the local capillary bed. Blood flow can increase sevenfold to eightfold as a result of vasodilation of the metarterioles and the relaxation of precapillary sphincters in response to an increased rate of metabolism. As the rate of metabolism increases in a tissue, vasodilator substances are produced in the extracellular fluid. Once produced, the vasodilator substances diffuse from the tissues supplied by the capillary to the area of the precapillary sphincter, the metarterioles, and the arterioles to cause vasodilation and relaxation of the precapillary sphincters (figure 21. Lack of oxygen and nutrients can also be important in regulating blood flow in tissues. For example, oxygen and nutrients are required to maintain vascular smooth muscle contraction. An increased rate of metabolism decreases the amount of oxygen and nutrients in the tissues. Smooth muscle cells of the precapillary sphincter relax in response to lower levels of oxygen and nutrients, resulting in vasodilation (figure 21.
References
- Fanucchi O, Ambrogi MC, Dini P, et al. Surgical treatment of non-small cell lung cancer in octogenarians. Interact Cardiovasc Thorac Surg 2011;12:749-53.
- Agostoni E, Zocchi L. Pleural liquid and its exchanges. Respir Physiol Neurobiol 2007;159:311-23.
- Yamashita M, Tanaka K, Matsuo T, et al. Cerebral dissecting aneurysms in patients with moyamoya disease: Report of two cases. J Neurosurg 1983;58:120.
- Sharretts JM, Simonds WF. Clinical and molecular genetics of parathyroid neoplasms. Best Pract Res Clin Endocrinol Metab 2010;24(3):491-502.
- Zomer A, van Rheenen J: Implications of extracellular vesicle (EV) transfer on cellular heterogeneity in cancer: what are the potential clinical ramifications? Cancer Res 76(8):2071n2075, 2016.
- Flanagan EP, O'Neill BP, Porter AB, et al. Primary intramedullary spinal cord lymphoma. Neurology 2011;77(8):784-91.
